All published articles of this journal are available on ScienceDirect.
Neurally Mediated Syncope in Children and Adolescents: An Updated Narrative Review
Abstract
Syncope presents a proportion of up to 25% among children and adolescents and accounts for 0.9% of emergency room visits. Its most frequent aetiology is neurally mediated syncope, which includes vasovagal syncope and orthostatic hypotension. Up to 70% of the paediatric population with reflex syncope is female. There are usually precipitating factors and prodromes. This mini-review will discuss the particularities of the clinical presentation, diagnosis, modified Calgary score, indications and accuracy of the head-up tilt test, classification, and pathophysiology in four sequential phases. Prognostic data and the non-pharmacological and pharmacological approaches will also be reviewed and differences regarding reflex syncope in adults will be discussed.
1. INTRODUCTION
The term syncope is derived from the Greek word “synkoptein” which means to interrupt, abbreviate, shorten [1, 2]. Neurally mediated syncope (NMS) is also called reflex syncope (vasovagal syncope) and may include orthostatic hypotension (OH). The term neurocardiogenic should be replaced by reflex syncope [3].
The proportion of syncope among children and adolescents is 25%, with a predominance between the ages of 8 and 18 years [4] and it is the reason for 0.9% of consultations in emergency departments [5]. A recent systematic review with 3,700 patients aged between 3 months and 21 years with syncope showed that the neurally mediated aetiology is the most common (52.2%), followed by postural orthostatic tachycardia syndrome (POTS) (13.1%). The cardiac cause was responsible for 4% of the cases of syncope and in 18.3% the cause was not identified [6].
Although NMS is a frequent condition in several settings in the paediatric population, the literature on this condition is not as comprehensive as it is for adults. Thus, this narrative mini-review aims to organize information about clinical aspects, pathophysiology, diagnosis and non-pharmacological and pharmacological approaches to syncope in children and adolescents.
2. METHODS
For this narrative mini-review, the electronic article search included the PubMed, EMBASE, and Google Scholar databases. The search terms used in relation to the topic of interest were: “vasovagal syncope’ (all fields) AND ‘children”, ‘vasovagal syncope’ AND ‘teenager’, OR ‘orthostatic intolerance’ AND 'children and adolescents'. Articles published from the year 2005 to November 2021 were included in the review, with priority for recently published articles.
3. CLINICAL PRESENTATION
Syncope is defined by sudden and transient loss of consciousness (short duration), with cerebral hypoperfusion and loss of postural tone, with immediate and spontaneous recovery, without the need for medication or cardiopulmonary resuscitation [3, 7]. The 6-8 s interruption of cerebral perfusion is enough for loss of consciousness [3].
In young people, there are usually precipitating factors for NMS, such as pain, emotional stress, dehydration, fear, medical or dental procedures or phobic disorders. The change from supine to sitting or standing position may precipitate NMS. The duration of the standing position of a precipitant can be >30 s and <2 to 3 min in vasovagal syncope; time is delayed OH, which is more than 3 min [3, 8]. There are situational triggers such as coughing, laughing, urination, defecation, swallowing, which are common in adults [7]. However, in 90% of cases of situational syncope triggered by cough in children, with facial congestion and changes in thoracic compliance, there is a history of asthma [9].
Prodrome can occur as pallor, sweating, warmth, abdominal pain, nausea, vomiting, and visual blurring [3, 7, 9]. During the loss of consciousness, the child or adolescent remains immobile, but in about 10% of cases, muscle movements can be observed without indicating a seizure [8]. On the other hand, 5-10% of episodes diagnosed as syncope can be seizure disorders [10, 11]. Fatigue can follow syncope and is more frequent in OH [12]. The presence of palpitations can help differentiate NMS from cardiac syncope as well as its occurrence on exertion or in the supine position [3, 7, 13].
There is a predominance of girls among the paediatric population with reflex syncope, reaching a proportion of 70% [11-15]. The syncope recurrence rate is between 24.7% and 38.5% [15, 16], with no gender preference, however it is higher in older patients [16].
4. DIAGNOSTIC AND MODIFIED CALGARY SCORE
The clinical history and physical examination performed with proficiency allow differentiating the cause of syncope in up to 60% of cases [3]. It should be ascertained whether there is a family history of premature sudden cardiac death. The physical examination should be comprehensive to identify signs of structural heart disease and the presence of orthostatic hypotension. An electrocardiogram (ECG) should also be done at the time of this initial assessment [3, 7, 14, 17]. Additional cardiovascular testing may be needed after this initial approach and will be discussed later in this article.
Syncope units in emergency departments can increase the accuracy of diagnosis and reduce costs in the paediatric population as well [18, 19].
In 2017, a document for practical evaluation/management of paediatric patients was published using the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) methodology [20]. To differentiate vasovagal syncope from other causes of syncope, the Modified Calgary score derived from the Calgary score for adults was developed. This score consists of seven diagnostic questions related to medical history with scores for positive answers to each question (Table 1). The score ranges from -14 to +6. A score <-3 suggests cardiac syncope. The sensitivity and specificity of scores below -2.5 are 96.3% and 72.7%, respectively, for differentiating cardiac syncope from POTS.
5. HEAD-UP TILT TEST
Head-up tilt test (TT) for the diagnosis of syncope has been debated. Its class of recommendation is IIa for reflex syncope, including OH and POTS. In patients with typical vasovagal syncope and emotional trigger, its positivity rate reaches 92%. However, TT can be positive in 47% of patients with cardiac syncope. Furthermore, its negative result does not exclude reflex syncope [3]. According to the Canadian Cardiovascular Society and Canadian Pediatric Cardiology Association Position Statement on the Approach to Syncope in the Pediatric Patient, TT has limited utility, but it can distinguish syncope from epilepsy in cases of recurrent episodes, especially if with video electroencephalogram monitoring [20].
| Questions | Points if Positive Answer |
| 1. Is there a history of bifascicular block, asystole, or supraventricular tachycardia? | -5 |
| 2. At times have bystanders noted that you turn blue during your faint? | -4 |
| 3. Did your syncope start when you were 5 years of age or younger? | -3 |
| 4. Do you remember anything about being unconscious? | -2 |
| 5. Do you feel faint with prolonged sitting or standing? | +1 |
| 6. Do you sweat before a faint? | +2 |
| 7. Do you feel faint with pain or in medical settings? | +3 |
According to the Consensus statement of the European Federation of Autonomic Societies [21], TT must be considered under the following conditions:
- when the clinical history, physical examination and ECG did not allow a definitive diagnosis of vasovagal syncope and POTS or for clarification of patients or family members or legal issues
- to differentiate syncope from seizure
- to differentiate vasovagal syncope from psychogenic causes
- for diagnosis of delayed OH, which occurs with more than 3 min of standing
- to guide patients to recognize prodromes and perform physical counter manoeuvres to avoid episodes of reflex syncope.
Its sensitivity and specificity vary by protocol, tilt angle, test duration and use of pharmacological provocation (nitroglycerin, isoproterenol). Sensitivity ranges from 21-72% and specificity can reach 99%, with a small decrease in the latter with the use of nitroglycerin (89%) [21].
To perform TT, fasting for 2-4 h is recommended. Rest time in the supine position should be at least 5 min (if TT without venepuncture) or 20 min (if TT with venepuncture). Tilt angle should be between 60 and 700, with tilt time <15 s. The duration of the passive phase should be 20-45 min. If the passive phase is negative, pharmacological provocation can be done with patient in the upright position. This phase should have a duration of 15-20 min, with nitroglycerin spray (single dose of 300-400 μg) or isoproterenol (dose of 1-3 μg/min, with the objective of increasing the heart rate by 20-25% in relation to the baseline heart rate) [3]. In the paediatric population, nitroglycerin can produce more prolonged vasovagal symptoms, with less sensitivity, and its routine use is not recommended [22, 23]. The flowchart of the head-up tilt test phases is shown in Fig. (1).
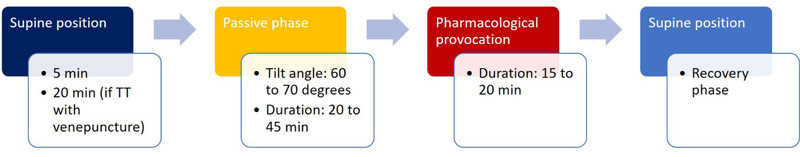
False-negative results may occur; however, the clinical history is paramount. The rate of false-positive results is comparable to other tests, such as the exercise stress test, but may indicate a propensity for syncope. There is the advantage of identifying the similarity between spontaneous symptoms related to syncope and those triggered in the laboratory during TT [24].
In the paediatric population, published studies have demonstrated the safety and usefulness of TT for the diagnosis and classification of syncope [11-13, 16], including children between 3-5 years of age [25]. Therefore, TT is considered a valuable diagnostic tool, after initial clinical approach with cost-effective appropriate, avoiding more expensive laboratory tests [24].
6. CLASSIFICATION OF NEURALLY MEDIATED SYNDROMES (NMS)
As per the Practical Instructions for the 2018 ESC Guidelines for the diagnosis and management of syncope [3], reflex syncope or NMS included vasovagal syncope, situational syncope, and OH. The classification of syncope is independent of age [26].
Vasovagal syncope may have the VASIS (VAsovagal Syncope International Study) classification modified [27], according to the TT response patterns.
- Type 1 mixed: there is drop in blood pressure (BP) and heart rate (HR). BP falls before HR falls. The ventricular rate does not fall to <40 beats/min.
- Type 2A, or cardioinhibitory without asystole: there is a drop in HR, with ventricular rate <40 beats/min for >10 s.
- Type 2B, cardioinhibition with asystole: there is asystole >3 s duration. BP decrease occurs simultaneously or before HR fall coincides with or precedes BP fall.
- Type 3 vasodepressor: there is decrease in BP below 80 mmHg, but HR does not fall >10%, from its peak, at the time of syncope.
In the paediatric population with a positive response to TT, the majority presented the vasodepressor response pattern (up to 77.4%), followed by the mixed response (up to 17.7%) and the cardioinhibitory response (up to 4.8%). The other response patterns are those of orthostatic intolerance, that is, OH or POTS [11, 25, 28].
Classical OH is defined as a sustained drop in systolic BP of at least 20 mmHg and/or diastolic BP of at least 10 mmHg from baseline or a drop in systolic BP to an absolute value <90 mmHg (especially in patients with systolic BP below 110 mmHg) within 3 min in active standing position. TT can be performed for diagnosis of delayed OH, which occurs with >3 min of standing [3].
OH can have factors such as dehydration or other forms of volume depletion, anaemia, drugs, and postprandial period. In neurogenic OH, there is no significant increase in HR (<10 bpm) due to dysfunction of the autonomic nervous system [3, 7] and it is less frequent in children [29]. Initial OH, in which there is a drop in BP (>40/20 mmHg) with the nadir 15 s, is the most common and can be confused with POTS. The clinical presentation is transient dizziness and there is usually no syncope [29, 30].
POTS is a set of signs and symptoms of chronic orthostatic intolerance (at least 6 months) that can infrequently present with syncope. Symptoms such as palpitations, blurred vision, dizziness occur when assuming the standing position, but other symptoms such as abdominal pain, nausea, diarrhoea, fatigue, sleep disturbances, migraine may occur unrelated to posture [3, 7, 8, 26, 31, 32]. A sustained increase in HR within 10 min of standing must be at least 40 bpm in those aged between 12-19 years and there is no drop in systolic BP >20 mmHg [3, 7, 8, 21]. Patients can have both conditions, POTS and reflex syncope, and therefore up to 30% of patients with POTS may experience syncope [33]. Therefore, POTS is not a cause of syncope, but it can be associated with it [26].

7. PATHOPHYSIOLOGY
Vasovagal syncope is a complex syndrome with individual variation, whose pathophysiology can also vary with age and is still not fully clarified. In the upright position, there are compensatory mechanisms of cerebral blood flow maintenance since 70% of the blood volume is below the level of the heart [29]. According to the haemodynamic changes obtained by non-invasive monitoring, there are four sequential phases in vasovagal syncope (Fig. 2) [34-36]. In phase 1, there is 500 to 1000 mL blood in the vessels below the diaphragm, which occurs with an 8-10 s nadir. This is the initial stabilization phase during TT. There is a 10-20% drop in cardiac output, as there is a reduction in central blood volume and the increase in HR is not compensatory, although tachycardia is more pronounced in children and young adults. Concomitantly, there is an increase in systemic vascular resistance of 40% due to sustained vasoconstriction of skeletal muscle arterioles and splanchnic arterioles. There is a transient increase in intra-abdominal pressure of an average magnitude of 43% in young people. Thus, mean BP is maintained by increasing diastolic BP. In phase 2, there is a drop in systolic and mean BP and a decrease in cerebral perfusion, with symptoms of presyncope. In young people, there is a loss of vasoconstrictor tone with vasodilation of the vessels of the upper limbs. At this phase, HR is higher in children and young adults by the action of the baroreflex, more exacerbated vagal tone, higher adrenaline levels and exaggerated nitric oxide activity. After an interval of 1-5 min, there is phase 3 in which there is a drop in BP. If the systolic BP reaches a value below 60 mmHg, syncope due to cerebral hypoperfusion occurs. There are differences in HR and cardiac output at this stage when comparing young people and older adults. The HR remains higher in young people, but the cardiac output is lower. The mechanism is uncertain, however the increase in adrenaline greater in young people may explain the vasodilator response of this substance via beta receptors [34]. Hypotension will result in vasodepressor response. Nonetheless, bradycardia or asystole may occur in young people during phase 3 due to vagal stimulation through a non-baroreflex pathway, that is, through the Bezold-Jarisch reflex, with a cardioinhibitory response. Phase 4 is the recovery phase, with increased venous return triggering the Frank-Starling mechanism and resulting increase in cardiac output and BP in the supine position.
The haemodynamic profile with low BP phenotype is described in vasovagal syncope. Young female patients, under 20 years of age, have lower systolic BP. This can be explained by the smaller body size and hormonal changes responsible for the accumulation of blood in the splanchnic territory, decreased plasma volume and vasodilation. However, these hormonal changes occur after puberty [37].
The participation of neurohormones in the pathophysiology of vasovagal syncope has not yet been determined, despite changes mainly in epinephrine, norepinephrine and arginine vasopressin with vasoactive actions during the phases of syncope in adults [38].
In the paediatric population, there is a report of an association of vitamin D deficiency with autonomic dysfunction (decreased parasympathetic action) and, therefore, with vasovagal syncope [39]. However, HR variability was not performed in the group of healthy children and adolescents for comparison with the group with syncope.
Regarding OH, its pathophysiology is explained by haemodynamic factors, if it is a non-neurogenic cause, such as drug-induced vasodilation or central hypovolaemia. Neurogenic OH is much less common in the paediatric population, but it can occur in conditions of primary or secondary dysautonomia, with inadequate release of norepinephrine from sympathetic neurons [29].
8. OTHER TESTS FOR NEURALLY REFLEX SYNDROMES
In addition to the tests already mentioned, such as ECG, TT, video electroencephalogram monitoring, other additional tests can be performed. Blood tests are not recommended routine, since they have low diagnostic value [7, 10, 21]. Catecholamine measurement can help in cases of neurogenic OH. CO2 expired records may show hypocapnia preceding reflex syncope or hyperventilation in the POTS [21]. Autonomic function tests can be used, since there is the involvement of the autonomic nervous system in the pathophysiology of NMS [3]. In cardiac syncope, imaging tests, such as echocardiography or others, cardiac monitoring (Holter, transtelephonic, loop, mobile telemetry), and electrophysiological study have their indications [3, 7, 10].
9. PROGNOSIS, INITIAL APPROACH AND TREATMENT OPTIONS
A study by Soteriades et al. including 727 participants from the original Framingham Heart Study and from the Framingham Offspring Study with a mean follow-up of 17 years, demonstrated that the prognosis of vasovagal syncope is benign, with no increase in mortality [40]. However, this study included participants over 20 years of age. Regardless of favourable prognosis, syncope can result in poor quality of life in the paediatric population, with scores similar to the quality of life of those with structural heart disease, advanced end-stage renal disease and asthma [41].
Despite the limited literature on evidence of non-pharmacological and pharmacological management of NMS in the paediatric population, the initial guidelines are class I recommendations. These initial guidelines to prevent recurrence of vasovagal syncope include clarification of its reflex mechanism, identification of precipitating factors, water (2 L/day) and salt intake and physical counterpressure manoeuvres [3, 7, 8, 10, 20]. The physical counterpressure manoeuvres should be performed during the onset of prodromes in order to avoid syncope. Examples of these manoeuvres are the movements of sitting, lying down, squatting, crossing legs, grasping hands and tensing them, pressing buttocks against each other, raising the lower limbs. Another isometric exercise such as squeezing an object of 6 cm in diameter in the dominant hand, may also be oriented as the child's age. Since these manoeuvres depend on the duration of the prodromes, they are class IIa of recommendation, according to some guidelines [7, 8], and are considered the first-line approach in patients with prodromal symptoms [42].
A recent meta-analysis of 5 randomized controlled trials in the paediatric population (average age from 11-13 years) with 233 in the intervention group and 175 in the control group demonstrated the efficacy of water and salt intake in preventing vasovagal syncope [43]. The dose per day of supplemental water was 500 ml and that of salt was 5.125 or 14.75 g. However, between a third to a half of the children had unsatisfactory results.
Orthostatic training can be effective in motivated young people [44] and the lower acceleration index can be a predictor of its effectiveness in children [45].
Pharmacological treatment with fludrocortisone and midodrine is Class IIb recommendations if there is failure of the initial steps [8]. In the multicentre randomized, placebo-controlled, double-blind, POST 2 (Prevention of Syncope Trial 2) study, there was a reduction in syncope recurrence after two weeks of fludrocortisone dose stabilization [46]. This efficacy in reducing the recurrence of syncope episodes was also observed in a retrospective observational study with 67 young people aged between 10 and 20 years, which compared the group of 28 patients using 0.1 mg once a day of fludrocortisone with the non-medication group [47].
Midodrine has been recommended for the paediatric population [20]. This alpha-agonist vasopressor was safe and effective for reducing the recurrence of vasovagal syncope in children at a dose of 2.5 mg twice a day [48], included in a randomized study with 26 patients [49].
There is a prospective, multicentre, randomized study (Prevention of Syncope Trial 5) in progress to verify whether beta-blocker therapy, such as metoprolol, would be effective in vasovagal syncope in patients over 40 years according to the results of a meta-analysis and an observational study [8]. In the paediatric population, there are studies with a small sample that verified the predictors of response to metoprolol, such as left ventricular ejection fraction and baroreflex sensitivity in the TT [50, 51]. Therefore, there is still no evidence from randomized, placebo-controlled trials for the use of beta-blockers in children and adolescents with recurrent vasovagal syncope.
Other pharmacological treatment options are emerging, but in young and adult subjects with vasovagal syncope. Reboxetine, sibutramine and atomoxetine, which act as inhibitors of the norepinephrine transporter and therefore prevent hypotension, may be promising drugs for the treatment of vasovagal syncope [52].
Syncope guidelines in the paediatric population also include pacemaker therapy and cardioneuroablation [20]. A study of 11 children with cardioinhibitory syncope refractory to conventional treatment, with a median age of 2.7 years, demonstrated abolition of syncope in 10 children with ventricle pacing and sensing (VVI mode) with hysteresis [53]. However, regarding the pacemaker, there is evidence of a reduction in the risk of reflex syncope due to asystole in patients aged at least 40 years, especially with closed loop system dual chamber pacing [54]. According to the Expert Consensus Statement on the indications of cardiovascular implantable electronic devices in paediatric patients [55], the indication for pacemaker implantation can be considered if there is an association between significant bradycardia or asystole and clinical syncope on more than one occasion in children or adolescents refractory to other conservative treatment options. This approach can improve the quality of life in selected paediatric patients.
Ablation of the right anterior ganglionated plexus as a primary target for cardioneuroablation has been performed to attenuate vagal activity in adult patients with vasovagal syncope [56-58]. Although, there are no data on this procedure in relation to the paediatric population. This procedure should be considered experimental even in adults over 40 years of age, since the studies included a small sample, there were complications, mainly of vascular access, and a mortality rate of 0.2% was reported [59].
| Initial approach - educational measures | - clarification of its reflex mechanism - identification of precipitating factors |
| Dietary measures and exercises | - water and salt intake - physical counterpressure maneuvers, if symptoms of prodromes - Orthostatic training |
| Pharmacological treatment | - fludrocortisone at a dose of 0.1 mg per day - midodrine at a dose of 2.5 mg twice a day |
| Options still lacking robust evidence in the pediatric population | - metoprolol - norepinephrine transporter inhibitors - pacemaker therapy - cardioneuroablation |
The main recommendations and pharmacological treatment options are listed in Table 2.
For patients with OH syncope, fluid and salt intakes are recommended and, in the case of neurogenic OH, midodrine can be used. Measures such as discontinuation of vasodilators, raising the headboard above 100 and approach to dysautonomia should be taken if they are involved with OH [3, 10].
CONCLUSION
Vasovagal syncope is the most frequent cause of NMS and its diagnosis can be made through clinical history and physical examination in up to 60% of cases. Modified Calgary score allows differentiation vasovagal syncope from other causes of syncope in the paediatric population. TT is considered a valuable diagnostic tool, after an initial clinical approach with adequate cost-benefit, and allows the modified VASIS classification. Although not fully elucidated, its pathophysiology included haemodynamic changes in four sequential phases and there may be neurohormonal participation. The initial guidelines to prevent its recurrence are educational measures and water and salt intake. Physical counterpressure manoeuvres are recommended in the prodrome period, which is frequent in young people. Fludrocortisone and midodrine can be used if initial guidelines fail. There is still no robust evidence in the paediatric population for pacemaker therapy and cardioneuroablation.
LIST OF ABBREVIATIONS
| BP | = Blood Pressure |
| ECG | = Electrocardiogram |
| HR | = Heart Rate |
| NMS | = Neurally Mediated Syncope |
| OH | = Orthostatic Hypotension |
| POTS | = Postural Orthostatic Tachycardia Syndrome. |
| TT | = Head-up tilt test |
| VASIS | = VAsovagal Syncope International Study |
CONSENT FOR PUBLICATION
Not applicable.
FUNDING
None.
CONFLICT OF INTEREST
The authors confirm that they have no conflicts of interest.
ACKNOWLEDGEMENTS
Declared none.


