All published articles of this journal are available on ScienceDirect.
Five-Year Outcomes of Heart Failure with Preserved Ejection Fraction
Abstract
Objective:
The aim of this study was to describe the clinical characteristics of patients that have had a heart failure with preserved ejection fraction (HF-pEF) and to identify the factors associated with 5-year mortality and readmission.
Methods:
A prospective cohort study was conducted of patients followed by the Heart Failure Unit of the Internal Medicine Department. Clinical characteristics and outcomes were collected. Univariate and multivariate analyses were performed in order to identify factors associated with 5-year mortality and readmission.
Results:
A total of 209 patients with HF-pEF were followed, 59.3% of these were women, with a mean age 79 years. The main etiology was hypertensive heart disease and a high level of comorbidity (chronic renal failure, hypertension and atrial fibrillation) was observed. The 5-year mortality was 55.5%; the related variables were anemia (hazard ratio [HR]=1.7; 95% confidence interval [CI]: 1.2-2.5), in patients being treated with statins (HR=0.7; 95%CI 0.5-0.9) and spironolactone (HR= 1.6; 95% CI: 1.1-2.3); 24.5% of patients had >2 admission in 5 years, with the main related factors being atrial fibrillation (HR=2.7; 95%CI: 1.4-5.5), anemia (HR=1.9; 95%CI:1.0-3.3) and were being treated with spironolactone (HR=2.1; 95%CI:1.2-3.7).
Conclusion:
Patients with HF-pEF are old and present a high level of comorbidity. Furthermore, they have a high 5-year mortality and readmission rate. The only factor associated with lower mortality was the treatment with statins. The use of spironolactone was associated with a higher mortality risk.
1. INTRODUCTION
Heart failure (HF) with preserved systolic function (HF-pEF) affects, on average, half of the patients with HF [1]. Its prevalence is increasing [2], which, in turn, represents an important increase in mortality, hospital admissions, and health costs [3]. Epidemiological characteristics of patients presenting with HF-pEF are different from those presented by the total number of patients with HF: they are older, with a greater proportion of women and obese people [4, 5].
However, the underlying physiopathological mechanism of HF-pEF is not clearly determined yet: while previously, a diastolic dysfunction of the left ventricle was suspected to be the cause, nowadays, new hypotheses are suggested, such as immunological alterations or the presence of a pro-inflammatory environment [6].
Although the same drugs used for HF with depressed function have been used, only a few studies have shown benefits derived from their use [7-10]. New strategies, such as targeted therapy and use of molecular paths in order to modify the physiopathology, have been suggested [11].
The 1-year mortality rate due to HF-pEF is about 20-30% in different studies [4, 12-14]. Several factors, such as anemia, diabetes mellitus, chronic kidney disease, or a large number of admissions, have been linked with a worse prognosis [4].
The aim of this study was to describe the clinical characteristics of patients with HF-pEF and to identify the factors associated with mortality and readmission in 5years of follow up.
2. MATERIALS AND METHODS
An observational study was conducted in a cohort of 338 patients who were followed in the Heart Failure Unit of the Department of Internal Medicine of the Santiago de Compostela University Teaching Hospital (Complexo Hospitalario Universitario de Santiago) (NW Spain). Patients were referred to this Unit from the cardiology, emergency, and internal medicine departments and primary health care. The patients were recruited from 1 January 2007 to 31 December 2011 [15].
After recruitment, it was found that all the patients were at least 50 years old. The diagnostic criteria used to include the patients were those published in the current guidelines [16, 17].
The patient information such as socio-demographic, clinical and analytical characteristics, along with the received treatment, were recorded, and then were followed for up to 5 years after the first consultation with a median follow-up of 4.73 (interquartile range [IQR]: 1.84-7.13) years. For this study, the patients that presented with HF-pEF (total cohort = 209 patients) were selected.
Left ventricular ejection fraction (LVEF) was measured using the Teichholz formula [18]. As preserved ejection fraction, a LVEF≥50%, was considered and as reduced ejection fraction, a LVEF<50%, was considered [16].
N Terminal pro-B-type natriuretic peptide (NT-proBNP) was measured with a commercial immunoassay (Cobas Elecsys e411, Roche), which employs two polyclonal antibodies directed at residues 1-21 and 39-50 of the NT-proBNP molecule.
Hypertension was defined as a blood pressure ≥140/90 mmHg or patients treated with antihypertensive treatment. Chronic kidney disease (CKD) was defined as a glomerular filtration rate <60 ml/min/1.73m2 using the 4-variable Modification of Diet in Renal Disease formula [19]. We considered as anemia a hemoglobin level <12 g/dl, iron deficiency as ferritin<12 ng/ml, vitamin B12 deficiency when vitamin B12 level was <200 pg/ml, folate deficiency when folate level was <5ng/ml and hypercholesterolemia when low density lipoprotein cholesterol (LDL-C) level was >130 mg/dl. Diabetes was defined as an abnormal glucose metabolism (hemoglobin A1c ≥6.5%, fasting plasma glucose ≥126 mg/dl, random venous plasma glucose ≥200 mg/dl in a patient with classic symptoms of hyperglycemia or plasma glucose ≥200 mg/dl measured 2h after a glucose load of 1.75 g/kg (maximum dose of 75 g) in an oral glucose tolerance test). Those patients who smoked tobacco regularly were considered as smokers and alcohol abusers were the patients who consumed >80 g alcohol/day.
HF etiology was classified as: ischemic, when the patient was diagnosed with ischemic Heart Disease (IHD); valvular, when the patient had at least a moderate valvulopathy without IHD; cor pulmonale, when there was a right heart failure without left ventricular dysfunction; and hypertensive, when the patient had hypertension without another cause of HF.
The study protocol was reviewed and approved by the Galician Clinical Research Ethics Committee (Reference number 2012/086) and conformed to the Helsinki Declaration. All data analyzed were anonymized.
2.1. Statistical Analysis
A descriptive analysis was performed by calculating qualitative variable rates plus mean and standard deviation. Either the Chi-squared test or Fisher’s exact test (when the expected frequency value is <5) was used to compare qualitative variables and Student’s t test was used for quantitative variables. Kaplan-Meyer curves were used to evaluate the factors related to mortality and readmission and a multivariate analysis was performed using a Cox-regression. A two-sided p<0.05 was regarded as significant. All analyses were performed using the SPSS v. 22.0 software package (SPSS Inc., Chicago, IL, USA).
3. RESULTS
During the period of study (up to 5 years), a total of 209 patients with HF-pEF, out of which, 59.3% were women, with a mean age of 79.0 years (standard deviation=7.2) were followed.
The main comorbid conditions were CKD (93.3%), hypertension (82.8%), atrial fibrillation (56.9%), and hypercholesterolemia (49.3%).
The most frequent etiology was hypertensive heart disease (64.1%), followed by ischemic disease (11.5%) and valvular disease (10.0%).
When the study started, the median of hemoglobin was 12.6 g/dl (IQR: 11.1-13.9), the median of the glomerular filtrate was 54.7 ml/min/1.73m2 (IQR: 40.0-67.3) and the median of NT-proBNP levels was 1368 pg/ml (IQR: 488-3302). The median of systolic blood pressure was 140 mmHg (IQR: 120-150) and the median of diastolic blood pressure was 75 mmHg (IQR: 65-80).
The most commonly used treatments were loop diuretics (90.9%) (median dose of furosemide:40 mg, IQR: 20-60 mg), statins (64.1%), and angiotensin receptor blockers (ARBs) (59.8%). Angiotensin-Converting Enzyme (ACE) inhibitors were used in 53.6% of the patients, less than beta-blockers (54.1%), and antiplatelet drugs (54.1%), as can be seen in Table 1.
| Characteristic | Result |
| Age (year, mean ± SD) | 79.0 ± 7.2 |
| Age ≥ 70 years, n (%) | 186 (89.0) |
|
Gender, n (%) Male Female |
85 (40.7) 124 (59.3) |
|
Comorbid conditions, n (%) CKD Hypertension Atrial fibrillation Hypercholesterolemia Anemia Iron deficiency Folate deficiency Vitamin B12 deficiency Diabetes mellitus COPD Smoking Alcohol abusers |
195 (93.3) 173 (82.8) 119 (56.9) 103 (49.3) 83 (39.7) 10 (4.8) 17 (8.1) 4 (1.9) 74 (35.4) 61 (29.2) 43 (20.6) 23 (11.0) |
|
Etiology, n (%) Hypertensive Ischemic Valvular Cor pulmonale Others |
134 (64.1) 24 (11.5) 21 (10.0) 16 (7.7) 14 (6.7) |
|
NYHA class, n (%) I II III IV |
58 (27.8) 86 (41.1) 61 (29.2) 2 (1.0) |
|
Treatment, n (%) Loop diuretics Statins Atorvastatin Pravastatin Simvastatin Rosuvastatin Others ARBs Beta-blockers Nebivolol Carvedilol Bisoprolol Atenolol Others Antiplatelet drugs ACE inhibitors Spironolactone Anticoagulants Calcium antagonists Nitroglycerin Digoxin Thiazide diuretics |
190 (90.9) 134 (64.1) 97 (72.4) 15 (11.2) 7 (5.2) 2 (1.5) 13 (9.7) 125 (59.8) 113 (54.1) 72 (63.7) 21 (18.6) 12 (10.6) 5 (4.4) 3 (2.6) 113 (54.1) 112 (53.6) 98 (46.9) 94 (45.0) 81 (38.8) 73 (34.9) 71 (34.0) 56 (26.8) |
|
Admissions, n (%) 0 1 2 3 More than 3 |
95 (45.5) 49 (23.5) 21 (10.0) 9 (4.3) 28 (10.2) |
| Five-year mortality, n (%) | 116 (55.5) |
|
Cause of mortality, n (%) Cardiovascular disease Infection disease Cancer Respiratory disease Renal failure Unknown |
33 (28.4) 13 (11.2) 5 (4.3) 1 (0.9) 1 (0.9) 63 (54.3) |
| - | Five-year mortality | More than 2 admission in 5-year follow-up | |||||
| Variable | n(%) who died (116) | n (%)survivors(93) | p | n(%) readmitted (49) |
n (%) not readmitted (160) |
p | |
| Male sex | 53 (45.7%) | 32 (34.4%) | NS | 22 (44.9%) | 63 (39.4%) | NS | |
| Comorbid condition | - | - | - | - | - | - | |
|
CKD Hypertension Atrial fibrillation Hypercholesterolemia Anemia Iron deficiency Folate deficiency VitaminB12 deficiency Diabetes mellitus COPD Smoking Alcohol abusers |
111 (95.7%) 100 (86.2) 71 (61.2%) 23 (27.1%) 56 (48.3%) 5 (4.3%) 9 (7.8%) 0 (0.0%) 35 (30.2%) 39 (33.6%) 27 (23.5%) 13 (11.5%) |
84 (92.3%) 73 (78.5%) 48 (51.6%) 28 (37.3%) 27 (29.0%) 5 (5.4%) 8 (8.6%) 4 (4.3%) 39 (41.9%) 22 (23.7%) 16 (18.2%) 10 (11.4%) |
NS NS NS NS <0.01 NS NS 0.04 NS NS NS NS |
47 (95.5%) 38 (77.6%) 37 (75.5%) 12 (31.6%) 26 (53.1%) 1 (2.0%) 3 (6.1%) 1 (2.0%) 23 (46.9%) 15 (30.6%) 9 (19.1%) 7 (14.9%) |
148 (93.7%) 135 (84.4%) 82 (51.3%) 39 (32.0%) 57 (35.6%) 9 (5.6%) 14 (8.8%) 3 (1.9%) 51 (31.9%) 46 (28.8%) 34 (21.8%) 16 (10.4%) |
NS NS 0.03 NS 0.03 NS NS NS 0.06 NS NS NS |
|
| Cause | |||||||
|
Hypertensive Ischaemic Valvular Cor pulmonale |
72 (62.1%) 14 (12.1%) 15 (12.9%) 9 (7.8%) |
62 (66.7%) 10 (10.8%) 6 (6.5%) 7 (7.5%) |
NS NS NS NS |
28 (57.1%) 6 (12.2%) 6 (12.2%) 7 (14.3%) |
106 (66.3%) 118 (11.3%) 10 (6.3%) 14 (8.8%) |
NS NS NS NS |
|
| Treatment | |||||||
|
Loop diuretics Statins ARBs Beta-blockers Antiplatelet drugs ACE inhibitors Spironolactone Anticoagulants Calcium-channel blockers Nitroglycerin Thiazide diuretics Digoxin Amiodarone |
100 (90.1%) 66 (56.9%) 65 (58.0%) 58 (51.8%) 58 (50.0%) 58 (52.7%) 62 (55.4%) 52 (44.8%) 42 (37.5%) 48 (42.9%) 30 (26.8%) 41 (35.3%) 15 (12.9%) |
90 (96.8) 68 (73.1%) 60 (64.5%) 55 (59.1%) 55 (59.1%) 54 (58.1%) 36 (38.7%) 42 (45.2%) 39 (41.9%) 25 (26.9%) 26 (28.0%) 30 (32.3%) 6 (6.5%) |
0.06 0.01 NS NS NS NS 0.02 NS NS 0.02 NS NS NS |
47 (97.9%) 31 (63.3%) 32 (66.7%) 25 (52.1%) 21 (42.9%) 24 (52.2%) 30 (62.5%) 31 (63.3%) 26 (54.2%) 25 (52.1%) 15 (31.3%) 25 (51.0%) 5 (10.2%) |
143 (91.7%) 103 (64.4%) 93 (59.3%) 88 (56.1%) 92 (57.5%) 88 (61.1%) 68 (43.3%) 63 (39.4%) 55 (35.0%) 48 (30.6%) 41 (26.1%) 46 (28.8%) 16 (10.0%) |
NS NS NS NS NS NS 0.02 0.01 0.02 <0.01 NS 0.01 NS |
|
| More than 2 admissions | -35 (30.2%) | -14 (15.1%) | 0.01 | - | - | - | |
COPD: chronic obstructive pulmonary disease; CKD: chronic kidney disease
Other clinical characteristics are shown in Table 1.
The 5-year mortality rate was 55.5% (116 patients) and 24.5% (58) of the patients that had>2 admissions during 5 years. The main cause of mortality was cardiovascular disease (29.1%).
The main factors derived by the univariate analysis (p<0.05) associated with 5-year mortality and having had >2 admissions are shown in Table 2.
The Kaplan-Meier curves for mortality (Fig. 1) show that the factors related were anemia, previous admissions, as well as treatment with spironolactone, statins, nitroglycerin, or loop diuretics.
The Kaplan-Meier curves for >2 admissions (Fig. 2) show that the related factors were atrial fibrillation, anemia, being treated with spironolactone, calcium channel-blockers, nitroglycerin, digoxin, and anticoagulants.
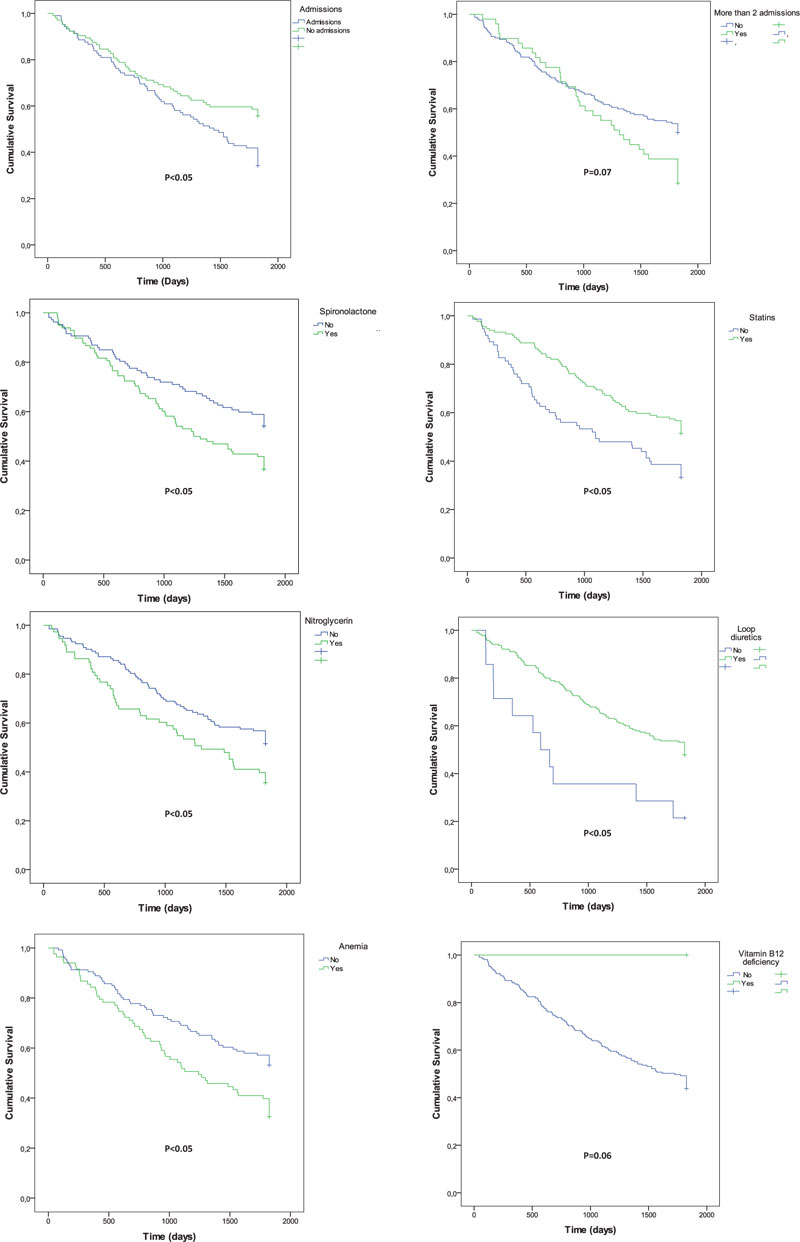

After a multivariate analysis, the variables associated with 5-year mortality were anemia (Hazard Ratio [HR]=1.7; 95% confidence interval [CI]: 1.2-2.5), being treated with statins (HR= 0.7; 95%CI 0.5-0.9) and spironolactone (HR=1.6; 95%CI: 1.1-2.3).
Nevertheless, the variables associated with having >2 admissions were atrial fibrillation (HR=2.7; 95%CI: 1.4-5.5), anemia (HR=1.9; 95%CI: 1.0-3.3), and being treated with spironolactone (HR=2.1; 95% CI: 1.2-3.7).
Patients treated with ARBs, ACE inhibitors, or beta-blockers showed no significant differences in mortality and readmission.
4. DISCUSSION
HF with preserved systolic function is a common disease that usually affects old patients and is associated with increased mortality and deterioration of well-being. Nowadays, no pharmacologic treatment has proven to result in a significant reduction in mortality and most of the treatments focus on addressing comorbidity to improve the well-being of the patient.
The patients with HF-pEF had a high 5-year mortality and readmission rate. To our knowledge, the majority of previous studies only evaluated 1-year outcomes [13, 14]; just 1 study had evaluated 5-year mortality and observed the rate of mortality >20% [4]. This reaffirms the importance of this disease and the need for efforts to improve outcomes.
Similar to previous studies, a high level of comorbidity, especially of CKD and hypertension [5, 13] was observed. Furthermore, like in other studies [4, 14], it was found that some comorbidities, like anemia, are correlated with greater mortality and readmission and some others, like atrial fibrillation, with more readmissions.
These clinical characteristics highlight the necessity to change the treatment in some cases. On the one hand, some comorbidities limit the response of treatment currently used in HF. For example, CKD reduces the response of diuretic treatments and restricts the use of drugs like ARBs, ACE inhibitors, or aldosterone antagonists.
On the other hand, as recommended by European Guidelines, a correct diagnosis and treatment of all comorbidities are needed to improve the symptoms and well-being and to reduce mortality and readmission rates [16].
As previous studies have found [7-9, 20-22], no change either in mortality and readmission rates was observed in patients treated with ARBs, ACE inhibitors, beta-blockers, or nitrates. Furthermore, more mortality and readmissions in patients treated with spironolactone were observed. Nevertheless, there are studies that have found some benefits with ACE inhibitors, ARBs, beta-blockers or aldosterone antagonists. In this context, the Charm-Preserved Trial [8] reported that the Candesartan reduced the admission rate for patients with HF-pEF and in the Perindopril Trial (PEP-CHF) [7], an improvement was found in symptoms and exercise capacity. Besides, the Study of Effects of Nebivolol Intervention on Outcomes and Rehospitalization in Seniors with heart failure (SENIORS) study [23, 24] found that the patients treated with nebivolol presented less mortality and readmission rates. Because of these reasons, the use of ARBs, ACE inhibitors and beta-blockers, especially to control hypertension, is considered reasonable in the present guidelines [17].
The spironolactone trials found contradictory results with regards to the improvement of symptoms. The Treatment of Preserved Cardiac Function Heart Failure With an Aldosterone Antagonist (TOPCAT) trial [25], which included 3445 patients with a median age of 68.7 years randomized to receive spironolactone or placebo with a mean follow-up of 3.3 years, found an improvement in the quality of life. On the other hand, the Aldosterone Receptor Blockade in Diastolic Heart Failure (Aldo-DHF)Trial [26], which included 422 patients with a median age of 67.0 years randomized to receive spironolactone or placebo with a mean follow-up was 11.6 months, did not find an improvement of the symptoms in these patients. The worst results in mortality and readmission with spironolactone can be explained because of a greater prevalence of CKD in the patients compared with the above mentioned studies [25, 26]. Nevertheless, since there is no clear evidence regarding the use of this treatment in HF-pEF, possibly we should consider avoiding aldosterone antagonists in these patients, especially if they present comorbidities, such as CKD.
To our knowledge, only 1 study evaluated the use of nitrates in HF-pEF [27] and did not find any change in symptoms with this treatment, and therefore, its use is not currently recommended [17].
Only the use of statin was associated with less mortality. To our knowledge, there are no previous studies that analyzed these treatments. Statins have previously been studied in HF with reduced ejection fraction and changes in outcomes have not been observed [28, 29]. Nevertheless, it is necessary to conduct more investigations to evaluate the outcomes in HF-pEF.
Calcium-channel blockers are not recommended to treat patients with HF with reduced ejection fraction [30, 31] and possibly they should also be avoided in HF-pEF, until future studies prove their safety.
The main limitations of the present study are its observational design and that it was conducted in a single center with a limited number of patients. However, the results are similar to those obtained by others. Moreover, not only patients admitted to the hospital but also ambulatory patients referred from primary health care were included. This provides a better vision of the general problem [4, 13, 14]. Furthermore, the study includes only patients followed by the Department of Internal Medicine, excluding the patients followed by the Cardiology Department. Probably because of this, the patients are older and with more comorbidity than in other studies [13, 14].
CONCLUSION
The HP-pEF is a common disease that usually affects old patients with a high number of comorbidities, inducing high mortality, high readmission rates, and deterioration of well-being.
The only factor associated with lower mortality was treatment with statins. On the other hand, spironolactone was associated with higher mortality and readmission rates.
It is necessary to continue studying this area to identify which factors can be modified in order to improve outcomes and well-being of the patients.
LIST OF ABBREVIATIONS
| ACE | = Angiotensin-Converting Enzyme; |
| Aldo-DHF | = Aldosterone Receptor Blockade in Diastolic Heart Failure; |
| ARBs | = Angiotensin receptor blockers; |
| CI | = Confidence Interval; |
| CKD | = Chronic kidney disease; |
| COPD | = Chronic Obstructive Pulmonary Disease; |
| HF | = Heart Failure; |
| HF-pEF | = Heart failure with Preserved Ejection Fraction; HR: Hazard ratio; |
| IHD | = Ischemic Heart Disease; IQR: interquartile range; |
| LVEF | = Left Ventricular Ejection Fraction; |
| NP-proBNP | = N Terminal pro-B-type natriuretic peptide; |
| NYHA | = New York Heart Association; |
| PEP-CHF | = Perindopril Trial; |
| SENIORS | = Study of Effects of Nebivolol Intervention on Outcomes and Rehospitalization in Seniors With Heart Failure; |
| TOPCAT | = Treatment of Preserved Cardiac Function Heart Failure With an Aldosterone Antagonist |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study protocol was reviewed and approved by the Galician Clinical Research Ethics, Committee, Spain (reference number 2012/086)
HUMAN AND ANIMAL RIGHTS
No animals were used in this research. All human research procedures were followed in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
Informed consent was obtained from all participants.
AVAILABILITY OF DATA AND MATERIAL
The data sets analyzed during the current study are available from the corresponding author [CM] upon request.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


