All published articles of this journal are available on ScienceDirect.
Atrial Fibrillation Recurrence Prevention after Electrical Cardioversion in High-Risk Patients – Benefits of Non-Antiarrhythmic Drugs
Abstract
Background:
Recurrence prevention after Atrial Fibrillation (AF) termination by Eelectrical Cardioversion (ECV) remains challenging. Increasing attention is paid to pathophysiological effects of non-Antiarrhythmic Drugs (non-AADs), nevertheless, with heterogeneous results.
Objective:
We evaluated the potential benefits of different non-AADs as adjunctive therapy to Antiarrhythmic Drugs (AADs) for AF recurrence prevention after sinus rhythm restoration by ECV in high-risk patients.
Methods:
The study was conducted among high-risk AF patients after successful ECV. Prescription of class IC or class III AAD was required. Data were acquired in a face-to-face baseline interview and 1-, 3-, 6-, 9-, 12-month follow-up interviews.
Results:
113 patients were included. Total AF recurrence rate reached 48.7%. Angiotensin-Converting Enzyme Inhibitor (ACEI) or angiotensin receptor blocker (ARB) intake, compared with non-use, demonstrated AF recurrence rate reduction by 8.5% (46.3 vs. 54.8%), with odds ratio (OR) reduced by 28.9% (OR 0.711, 95% confidence interval (CI) 0.310-1.631, p = 0.420). Among mineralocorticoid receptor antagonist (MRA) users, AF recurrence rate was reduced by 25.1% (29.6 vs. 54.7%) and OR by 65.1% (OR 0.349, 95%CI 0.138-0.884, p = 0.023). Present statin therapy reduced AF recurrence rate by 4.2% (46.8 vs. 51.0%) and OR by 15.5% (OR 0.845, 95%CI 0.402-1.774, p = 0.656). Diuretic use showed reduction of AF recurrence rate by 10.2% (41.7 vs. 51.9%) and OR by 33.9% (OR 0.661, 95%CI 0.297-1.469, p = 0.308).
Conclusion:
Non-AADs demonstrated practical benefits as adjunctive therapy to AADs for AF recurrence prevention after ECV in high-risk patients, with statistically significant results established for concomitant MRA intake.
1. INTRODUCTION
Atrial Fibrillation (AF) is the most prevalent arrhythmia, considered to be a worldwide epidemic and also associated with increased morbidity and mortality [1-3]. Rhythm control, aimed at restoration and maintenance of sinus rhythm [4], is one of the cornerstones of arrhythmia management [5], especially if AF is symptomatic and poorly tolerated [6, 7]. Electrical Cardioversion (ECV) is a quick and effective method [8], terminating AF in > 90% of the cases [9]. Antiarrhythmic drug (AAD) therapy is a common practice with established effectiveness for post-cardioversion sinus rhythm maintenance [10, 11]. Class IC and class III medications are widely used [10, 12], and class II antiarrhythmic properties have also demonstrated some clinically significant efficacy [11, 13, 14].
Despite treatment advances, arrhythmia recurrence prevention remains a therapeutic challenge [14, 15]. Furthermore, higher CHA2DS2-VASc (congestive heart failure, hypertension, age ≥ 75 years (doubled), diabetes mellitus, stroke (doubled), vascular disease, age 65-74 years, sex category (female) [8]) score (at least 2) values have been linked to modest success, which can be attributed to the accumulation of pathogenetic contribution of the specific patient characteristics and comorbidities that form the basis of calculation [16, 17]. Increasing attention is, therefore, being paid to underlying pathophysiological background [5, 18], and effects of non-Antiarrhythmic Drugs (non-AADs), referred to as upstream therapies, evoke rising interest [8, 19]. Recently, an updated classification of AADs has been proposed, where Angiotensin-Converting Enzyme Inhibitors (ACEIs), Angiotensin Receptor Blockers (ARBs), omega-3 fatty acids, and statins were included as class VII AADs, based on the reduction of arrhythmic tendency through modifying electrophysiological and structural remodeling [20]. Besides ACEIs and ARBs, Mineralocorticoid Receptor Antagonists (MRAs) exerting an inhibitory effect on renin-angiotensin-aldosterone system (RAAS), have a substantiated role [21, 22]. Diuretics have relatively less evidence, however, a potential impact through the reduction of atrial size and wall stress is also noteworthy [23]. Nevertheless, results obtained in various studies are comparatively heterogeneous, the role of non-AADs remaining, to some extent, ambiguous [24-26].
The aim of this study was to evaluate the potential benefits of different non-AADs (ACEIs/ARBs, MRAs, diuretics and statins) as adjunctive therapy to AADs for AF recurrence prevention after sinus rhythm restoration by ECV in high-risk patients.
2. MATERIALS AND METHODS
2.1. Ethical Approval
The study was conducted, and data were acquired on the basis of ethical approval from the Research Ethics Committee of Riga Stradins University Nr.6-3/16 (September 27, 2018). All patients signed informed consent.
2.2. Patient Inclusion and ECV Procedure
This prospective study was conducted among AF patients undergoing ECV in the Latvian Centre of Cardiology, Pauls Stradins Clinical University Hospital, from October 2018 to June 2019. Inclusion criteria were:
- Age ≥ 18 years
- Non-valvular AF: mainly excluding patients that have either rheumatic valvular disease or mechanical heart valves, according to the European Society of Cardiology (ESC) Guidelines [27]
- High-risk AF: defined by CHA2DS2-VASc score 2 or higher for men and 3 or higher for women, as specified in the ESC Guidelines [27], thus adapting a factor associated with higher susceptibility to AF recurrence after ECV [16, 17].
- Successful AF conversion to sinus rhythm by ECV.
- Prescription of a class IC or class III AAD, representing antiarrhythmic mechanisms of action with established effectiveness [10, 12].
Concomitant beta-blocker intake was neither mandatory nor restricted, but it was taken into account when analysing the outcomes, as class II antiarrhythmic properties can have an impact, facilitating sinus rhythm maintenance [11, 13, 14].
ECV procedure was conducted according to a standard developed and accepted in the Latvian Centre of Cardiology, Pauls Stradins Clinical University Hospital. Standardized Protocol of Elective ECV, adapted for performing the procedure in the hospital, and developed in accordance with recommendations derived from guidelines and consensus documents accepted in the ESC [9, 27, 28]:
- Clinical examination (performed to rule out causes of secondary AF and investigate potential significant structural heart disease):
- Medical history
- Physical examination
- General laboratory tests (including International Normalized Ratio (INR) tests in the last 3 weeks with INR in the range 2.0-3.0, D-dimers just before cardioversion, natriuretic peptides)
- Echocardiographic examination
- Outpatient pharmacological pretreatment:
- Anticoagulant administration: direct oral anticoagulants (dabigatran, rivaroxaban or edoxaban) are started at least 3 weeks before ECV; for vitamin K antagonists (warfarin), 3-week time period is counted from the stable achievement of INR target (at least 2.0)
- AADs: initiated 7-10 days before ECV (preference is given to class IC AADs if there are no significant contraindications; another option is amiodarone or sotalol)
- Used rate control therapy: beta-blockers or non-dihydropyridine class calcium channel blockers (verapamil or diltiazem)
- Digoxin should be discontinued at least 7 days before ECV
- RAAS-modulating drugs (ACEIs, ARBs, MRAs), statins, metabolic therapy, and other necessary agents are not discontinued.
- ECV procedure:
- If a patient has prolonged AF, high risk of stroke/systemic embolism according to the CHA2DS2-VASc score, an unclear history of anticoagulant use, and high risk for thrombus in left atrial appendage, transesophageal echocardiography is performed prior to ECV
- Standard equipment: biphasic defibrillator LifePak, self-adhesive pads in the modified anteroposterior position (left subscapular and right parasternal position) or classical anterior-posterior position;
- Intravenous sedation: propofol solution, 0.1-0.15 mg/kg/min intravenously for 3-5 min, administered as slow infusion or slow injection and titrated to desired clinical effect while monitoring cardiorespiratory functions;
- Protocol for increasing Direct Current (DC) shock energy: 5 DC shocks in standard, starting with 200 J, followed by 300 J, 360 J two times and 360 J with chest compression;
- An acute successful outcome is defined as sinus rhythm maintenance for at least 15 min after ECV.
- In case of unsuccessful cardioversion, the rate control strategy is considered, conceptual change of pharmacotherapy is initiated, or possible catheter ablation is planned if no contraindications.
- Outpatient follow-up:
- After ECV, the patient is monitored by their general practitioner in collaboration with an outpatient cardiologist;
- Typical follow-up involves clinical and electrocardiographic examination, Holter monitoring in case of potential short episodes of recurrence, control of medication intake, including anticoagulant therapy (continued for 4 weeks after ECV in patients with no need in long-term anticoagulation, while lasting like a permanent long-term therapy in high-risk AF patients, defined based on CHA2DS2-VASc score values of at least 2 in men and 3 in women) and use of AADs.
2.3. Data Acquisition
Baseline data was acquired in a face-to-face interview, according to a data collection protocol, obtaining information on demographics, medical history (including data available from previous investigations) and medication intake. Subsequently, 1-, 3-, 6-, 9- and 12-month follow-up telephone interviews were conducted, focusing on intake of medications, patient compliance, medication persistence and sinus rhythm maintenance. The first experienced post-cardioversion episode of AF was considered a recurrence, and subsequent heart rhythm changes were not taken into consideration. Adherence to pharmacotherapy within 12 months, or at least until AF recurrence was required, when selecting data for further statistical evaluation.
2.4. Statistical Analysis
Data analysis was carried out using Microsoft Excel and IBM SPSS Statistics software version 26.0 (Armonk, NY: IBM Corp). Descriptive statistics were used, presenting quantitative normally distributed data (according to the Kolmogorov-Smirnov test for normality) as mean and Standard Deviation (SD) and non-normally distributed data as the median and interquartile range (IQR), categorical and ordinal values as median and IQR and calculating frequencies for categorical nominal values. The total AF recurrence rate, as well as recurrence rates and their differences among various non-AAD users and patients not having particular non-AADs in therapy, were determined. By evaluating the effectiveness of non-AADs, Odds Ratios (ORs) were calculated based on contingency tables, and 95% Confidence Intervals (CIs) were used for data representation. Pearson’s chi-square test was used to determine statistical significance comparing medication intake between groups and estimate the association between medication intake and AF recurrence. A 95% CI level with the corresponding two-tailed p-value < 0.05 defining statistical significance was set.
3. RESULTS
3.1. Patient Characteristics
The total number of participants was 113. The mean age of patients was 65.5 (SD 9.4) years. Among the study participants, 57.5% (n = 65) were men with the median CHA2DS2-VASc score value 3 (IQR = 2-4), and 42.5% (n = 48) women, with the median for CHA2DS2-VASc score 4 (IQR 3-5). Regarding represented types of AF, 8.8% (n = 10) of patients had paroxysmal AF, 85.0% (n = 96) persistent AF, and 6.2% (n = 7) longstanding persistent AF. The median for the duration of AF episode till ECV was 8 (IQR = 4-17) weeks. The median for total AF history among patients was 17 (IQR = 2.5-48) months. Baseline characteristics of study participants are summarized in Table 1.
| Parameters | Values | |
|---|---|---|
| Total number of patients | 113 | |
| Age, years, mean (SD) | 65.5 (SD 9.4) | |
| Sex | Men Women |
57.5% (n = 65) 42.5% (n = 48) |
| CHA2DS2-VASc score | Men – median (IQR) Women – median (IQR) |
3 (IQR 2-4) 4 (IQR 3-5) |
| AF type | Paroxysmal Persistent Longstanding persistent |
8.8% (n = 10) 85.0% (n = 96) 6.2% (n = 7) |
| AF episode duration till ECV in weeks, median (IQR) | 8 (IQR = 4-17) | |
| AF history in months, median (IQR) | 17 (IQR = 2.5-48) | |
| AAD | ACEI/ARB | MRA | Statin | Diuretic | ||||
|---|---|---|---|---|---|---|---|---|
| - | Users | Non-users | Users | Non-users | Users | Non-users | Users | Non-users |
| 72.6% (n = 82) |
27.4% (n = 31) |
23.9% (n = 27) |
76.1% (n = 86) |
54.9% (n = 62) |
45.1% (n = 51) |
31.9% (n = 36) |
68.1% (n = 77) |
|
| Class IC | 32.9% (n = 27) |
38.7% (n = 12) |
33.3% (n = 9) |
34.9% (n = 30) |
35.5% (n = 22) |
33.3% (n = 17) |
27.8% (n = 10) |
37.7% (n = 29) |
| p = 0.564 | p = 0.882 | p = 0.811 | p = 0.303 | |||||
| Concomitant beta-blocker | 96.3% (n = 26) |
100% (n = 12) |
100.0% (n = 9) |
96.7% (n = 29) |
95.5% (n = 21) |
100.0% (n = 17) |
90.0% (n = 9) |
100.0% (n = 29) |
| p = 0.499 | p = 0.579 | p = 0.373 | p = 0.084 | |||||
| Class III | 67.1% (n = 55) |
61.3% (n = 19) |
66.7% (n = 18) |
65.1% (n = 56) |
64.5% (n = 40) |
66.7% (n = 34) |
72.2% (n = 26) |
62.3% (n = 48) |
| p = 0.564 | p = 0.882 | p = 0.811 | p = 0.303 | |||||
| Concomitant beta-blocker | 76.4% (n = 42) |
68.4% (n = 13) |
77.8% (n = 14) |
73.2% (n = 41) |
77.5% (n = 31) |
70.6% (n = 24) |
76.9% (n = 20) |
72.9% (n = 35) |
| p = 0.494 | p = 0.700 | p = 0.498 | p = 0.706 | |||||
3.2. Medication Intake
An ACEI/ARB was used by 72.6% (n = 82) of the patients and 23.9% (n = 27) of the study participants were taking MRAs. Statin therapy was present in 54.9% (n = 62) of cases, and 31.9% (n = 36) of the patients were on diuretics.
For included patients to be in more equivalent positions and thus ensuring greater objectivity in evaluating the effect of non-AADs, use of AADs was taken into account, and therefore use of a class IC or class III AAD was an inclusion criterion. 34.5% (n = 39) of patients were taking a class IC antiarrhythmic medication, represented by the use of ethacizine in 25.7% (n = 29) of the cases and propafenone among 8.8% (n = 10) of study participants. A class III AAD was used by 65.5% (n = 74) of the participants, where 56.6% (n = 64) were amiodarone users and 8.8% (n=10) were sotalol users. By analysing the distribution of class IC and class III AAD intake, no statistical significance was established between users and non-users of different non-AADs. Regarding beta-blocker intake, it was not restricted, but it was taken into consideration. Comparing rates of concomitant beta-blocker intake among class IC or class III AAD users, no statistical significance was established with all non-AAD user and non-user groups, as well. The corresponding data is presented in Table 2.
3.3. Effect of Non-AAD Intake on AF Recurrence Prevention
The total AF recurrence rate reached 48.7%, with the median for time from the baseline ECV procedure to the first AF recurrence 5 (IQR 1-19.5) weeks. For patients on ACEI/ARB therapy, the AF recurrence rate was 46.3%, whereas, among non-users, it was 54.8%, demonstrating that ACEI/ARB intake, compared with non-use, reduced AF recurrence rate by 8.5%. MRA use showed a reduced AF recurrence rate of 25.1%, with AF recurrence rates 29.6% and 54.7% among users and non-users, respectively. In patients on statin therapy, the AF recurrence rate reached 46.8% and was 51.0% among non-users, with AF recurrence rate reduction by 4.2%. Finally, diuretic use reduced AF recurrence by 10.2%, with an arrhythmia recurrence rate of 41.7% among diuretic users and 51.9% in patients who did not take a diuretic. Graphical representation of data is shown in Fig. (1).
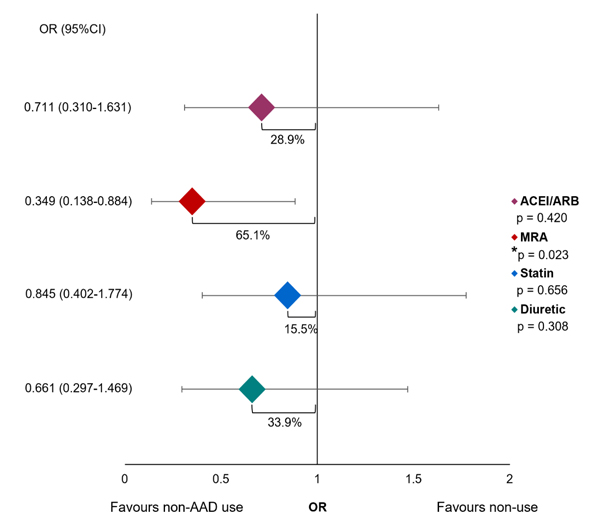
AF: atrial fibrillation, non-AADs: non-antiarrhythmic drugs, ACEI/ARB: angiotensin-converting enzyme inhibitor/ angiotensin receptor blocker, MRA: mineralocorticoid receptor antagonist ACEI/ARB intake demonstrated a reduction in OR for AF recurrence by 28.9% (OR 0.711, 95% CI 0.310-1.631), but this difference did not achieve significance (p = 0.420). Among MRA users, the OR for arrhythmia recurrence was reduced by 65.1% (OR 0.349, 95% CI 0.138-0.884) with a statistically significant difference (p = 0.023). With statin therapy, the OR for AF recurrence was reduced by 15.5% (OR 0.845, 95% CI 0.402-1.774), with no statistically significant difference (p = 0.656). Diuretic use showed a reduction of the OR for AF recurrence by 33.9% (OR 0.661, 95% CI 0.297-1.469), but without statistically significant difference (p = 0.308). These outcomes are summarized in Fig. (2).
Non-AAD: non-Antiarrhythmic Drug, ACEI/ARB: Aangiotensin-Converting Enzyme Inhibitor/Angiotensin Receptor Blocker, MRA: Mineralocorticoid Receptor Antagonist, OR: Odds Ratio, CI: Confidence Interval

4. DISCUSSION
Non-AADs, also known as upstream agents, come into focus in research regarding AF and arrhythmia recurrence prevention, yet, to some extent, remaining in a debatable position [5, 29]. The concept of this treatment approach is based on the potential of these medications to interfere with the pathophysiological mechanisms that determine remodeling associated with arrhythmogenic substrate development, AF susceptibility, and perpetuation [30]. Important proarrhythmic alterations are linked to structural changes [31], including developments, such as atrial fibrosis [32], inflammatory processes [33], atrial enlargement [34], as well as electric remodeling [35]. Based on these considerations, the potential benefits of non-AAD therapy for AF recurrence prevention after sinus rhythm restoration by ECV in high-risk patients were evaluated in this prospective study, including ACEIs/ARBs, MRAs, statins, and diuretics.
Analysing the characteristics of the patient population, there are certain factors to emphasize regarding outcome evaluation. Inclusion of high-risk AF patients per se, reinforced with an estimated median of 3 (IQR 2-4) among men and 4 (IQR 3-5) among women, is representative of accumulation of relapse-predisposing factors, including demographic character- istics and comorbidities [15, 17]. Persistent AF type was mostly represented, indicating results mainly attributable to this group. Complementary data regarding overall AF history is also of importance, with an estimated median value of 17 (IQR 2.5-48) months, demonstrating a considerable time period since AF diagnosis. This information underlines association with the progressive remodeling process and, to a certain extent, already present electrical and structural alterations at the time of baseline cardioversion procedure. Advanced progression is associated with a poorer sinus rhythm maintenance prognosis. On the other hand, the pharmacological inhibitory effect of indirect antiarrhythmic action on underlying pathophy- siological processes could be particularly beneficial, interrupting arrhythmia self-maintenance and reducing treatment resistance [13, 32].
In this study, the effects of non-AADs were viewed from the perspective of adjunctive therapy to antiarrhythmic medications since AADs have an established role in promoting post-cardioversion sinus rhythm maintenance [36]. Therefore, class IC or class III AAD intake was a requirement for inclusion in the study, representing medications commonly used in practice in this context [10]. There is certain evidence that AAD choice does not have a significant impact on outcomes regarding AF relapse after ECV [37]; nevertheless, data suggesting the superiority of class III antiarrhythmic mechanism of action, particularly amiodarone, are also available [38, 39], which is important, taking into consideration that in this study, class III was represented by amiodarone with a considerable predominance, used by more than half of participants. Thereby, additionally, the distribution of AAD intake among different non-AAD users and non-users was evaluated, without any statistical significance determined. Beta-blocker intake was also taken into account since class II antiarrhythmic mechanism of action can also contribute to arrhythmia-free period prolongation [11, 24, 38]. Beta-blocker intake, concomitant to class IC and class III AADs, among users and non-users of different non-AADs, was evaluated, with no statistical significance established. Thus, the impact of particular non-AADs, potentially influencing sinus rhythm maintenance outcomes, was equivalent. Therefore, higher accuracy and validity, when evaluating and interpreting the contribution of investigated non-AADs to AF recurrence prevention, was provided.
The total AF recurrence rate, estimated in this study, was approximately 50%, indicating that almost half of the study participants experienced an arrhythmia relapse during the follow-up period, highlighting the importance of this issue and indicating that therapeutic improvements would be appropriate. Furthermore, the significance of AF relapse is underlined by early occurrence, with an estimated median for recurrence barely exceeding 1 month.
Regarding the evaluation of effects of investigated non-AADs and their impact on AF recurrence rates and the odds to experience arrhythmia relapse, all included medications demonstrated a trend towards more favourable outcomes when comparing users and non-users. Nevertheless, particularly demonstrated results and more detailed analysis for each non-AAD are worth discussing, provided below, including highlights of potential theoretical background and also in the context of available data.
An important target for non-AAD therapy is RAAS [40]. Activation and upregulation of RAAS have been linked to AF through several pathophysiological changes [41], associated with both electrophysiological alterations and structural remodeling [22]. The contribution of increased ACE expression and angiotensin II-mediated pathways has been described [26, 42] with analogously great importance of aldosterone-mediated processes in the pathogenesis of AF [30, 43, 44]. RAAS inhibition at different levels can be achieved with ACEIs and ARBs, as well as aldosterone antagonists [45], also referred to as MRAs [21]. In the context of AF recurrence prevention, in different studies, ACEI/ARB treatment has overall demonstrated benefits [46]; nevertheless, a comparative variability in data in different publications has also been noted [24, 47]. In the present study, ACEI/ARB therapy reduced the AF recurrence rate, but this effect was not statistically significant. MRA therapy has overall demonstrated a notable efficacy with a significant reduction of AF recurrence [21, 48], however, in this regard, research outcomes are also heterogeneous [49]. In the present study, MRAs demonstrated the greatest difference in AF recurrence rate and OR compared with their non-use, and this difference was statistically significant. Although ACEIs, ARBs and MRAs all exert an inhibitory effect on RAAS, the particular tendency observed in the present study could possibly be linked to clinical relevance and pathogenetic aspects of aldosterone effects within RAAS, also highlighting its important causative role in the context of AF and rhythm control resistance, including post-cardioversion sinus rhythm maintenance [43, 44, 50]. Therefore, more prominent efficacy could have been observed for direct pharmacological MRA blockade, significantly eliminating the adverse effects of aldosterone. Taking into consideration the supportive arguments and promising findings in our study in favour of MRA therapy, the next field of interest would be the patient profile suitable for receiving such treatment. Previously, studies regarding AF prevention and MRA therapy highlight patients with heart failure as a population that could benefit from this treatment option [21, 43]. Additionally, it has been suggested that a longer AF history and highly recurrent arrhythmia could define patient selection for MRA intake, however, usefulness and substantial positive effect for patients without heart failure and less advanced disease has been described [21]. Overall, conclusive answers regarding this question are still pending, therefore, there is a need of a direction for further research, with a greater focus on patient characteristics, intended to define the target population for MRA therapy for an indication associated with AF.
Statins, typically and primarily associated with lipid-lowering action [51], are also known for their pleiotropic effects, potentially underlying their antiarrhythmic efficacy [52], also facilitating the maintenance of sinus rhythm after ECV [53]. Nevertheless, the positive effect of statins resulting in coronary artery disease progression retardation should be mentioned since ischemia contributes to AF development [19]. Data supporting the significant use of statin therapy for AF relapse prevention is available [54], however, the conclusions are overall unconvincing [24]. In this study, statin therapy demonstrated a slight reduction in AF recurrence rate and OR, yet overall showing the least benefit when comparing to its non-use. Such observation could possibly be linked to the concept of changes of pathogenetic pathways with AF establishment and propagation, leading to the reduced significance of statin impact [53, 55]. In the present study, persistent AF was the most represented type, potentially supporting this explanation for the lack of noteworthy statin effect, determined in other post-cardioversion research [56].
Regarding the potential role of diuretics for post-cardioversion sinus rhythm maintenance, relatively limited data is available. Nevertheless, it should be noted that previous research has shown a positive impact of diuretic therapy regarding AF [23]. The expected efficacy of diuretic use has a theoretical background supportive of potential benefits. AF is associated with chronic atrial stretch and haemodynamic alterations, having a major contribution in promoting AF by induction of structural changes and altered electrical conduction [57-59]. In this study, diuretic use demonstrated, although not statistically significant, however a noticeable reduction of AF relapse rate and OR for AF recurrence.
The results of the present study are supportive of the importance of the integration of upstream therapies within rhythm control strategy [18, 60], including their valuable role for post-ECV sinus rhythm maintenance [19, 24], in particular for high-risk AF patients. Therefore, on the basis of these findings, supplementing the current level of evidence further expanded research should be encouraged. It would be relevant to conduct more detailed analyses of the potential benefits of different non-AAD intake among various patient subgroups with a focus on patient characteristics and specific comorbidities. Such investigations could help identify specific target patient populations for each medication group.
CONCLUSION
Non-AADs (ACEIs/ARBs, MRAs, diuretics, and statins) demonstrated a potential trend for added benefit as adjunctive therapy to AADs for AF recurrence prevention after sinus rhythm restoration by ECV in high-risk patients. However, the only statistically significant result was for concomitant MRA intake. Larger studies are needed in this field.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The study was conducted, and data were acquired on the basis of ethical approval from the Research Ethics Committee of Riga Stradins University with approval number Nr.6-3/16 (September 27, 2018).
HUMAN AND ANIMAL RIGHTS
No animals were used as the basis of this study. The human research procedures were conducted according to the Declaration of Helsinki principles.
CONSENT FOR PUBLICATION
All patients signed informed consent, confirming participation in the research.
AVAILABILITY OF DATA AND MATERIALS
Not applicable.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
We would like to express our gratitude to the hospital staff engaged in the research.


