All published articles of this journal are available on ScienceDirect.
Clinical Characteristics, Comorbidities, and Prognosis in Patients with Heart Failure with Unknown Ejection Fraction
Abstract
Background:
Heart Failure (HF) is a frequent cause of mortality and recurrent hospitalization. Although HF databases are assembled based on left ventricular (LV) ejection fraction, patients without LV ejection fraction determination are not further analyzed.
Objective:
The purpose of this study is to characterize patient attributes and outcomes in this group-HF with unknown Ejection Fraction (HFunEF).
Methods:
We queried the electronic medical record from a community-based university practice for patients with a HF diagnosis. We included patients with >60 days follow-up and had interpretable Doppler-echocardiograms. We recorded demographic, Doppler-echocardiographic, and outcome variables (up to 2083 days).
Results:
There were 820 patients: 269 with HF with preserved Ejection Fraction (HFpEF), 364 with HF with reduced Ejection Fraction (HFrEF), of which 231 had a LV ejection fraction=40-49% and 133 had a LV ejection fraction<40%, and 187 with HFunEF. As compared to patients with HFunEF, HFpEF patients were younger, had a higher coronary disease and hyperlipidemia prevalence. Patients with HFrEF had more prevalent coronary disease, myocardial infarction, and hyperlipidemia. Patients with HFunEF were more likely to be seen by non-cardiology providers. All-cause mortality (ACM) was greater in HFunEF patients than patients with HFpEF (Hazard Ratio (HR)=1.60 (1.16-2.29), p=0.004). Furthermore, HF readmission rates were lower in HFunEF as compared to HFpEF (HR=0.33 (0.27-0.54), p<0.0001) and HFrEF (HR=0.30 (0.028-0.50), p<0.0001).
Conclusion:
Patients with HFunEF have greater ACM and lower HF re-admission than other HF phenotypes. Adherence to core measures, including LV ejection fraction assessment, may improve outcomes in this cohort of patients.
1. INTRODUCTION
HF is the leading cause of morbidity, mortality and recurrent hospitalization. It is most common in the elder population accounting for 875,000 admissions each year [1] with prevalence increasing further with age. The prevalence of the HF in the elderly population has been increased due to improved survival following myocardial infarction and increased prevalence of co-morbidities including hypertension, diabetes, metabolic syndrome, valvular heart disease and coronary artery disease [2-4]. HF databases are usually assembled based on LV ejection fraction. Within the patient population, there may exist a group of patients (<20%) who do not have a LV ejection fraction determination and are not further analyzed or a previous assessment of LV function obtained several years in the past has been used [5-7]. There is sparse data available in the literature about patient characteristics and outcomes in this group of patients that we are defining as HFunEF. The purpose of this study is to characterize patient attributes and describe outcomes in this group as compared to patients with other HF phenotypes: HFpEF and HFrEF.
2. MATERIALS AND METHODS
This study received expedited approval by the Quillen College of Medicine Institutional Review Board. The Quillen College of Medicine clinical practice is contained in a single electronic medical record (Allscripts, Touchworks, Chicago, Ill) and was queried for HF diagnosis using ICD-9 codes (428.xx), HF admission, or HF on a problem list. There were 863 patients with HF that were identified. Only those patients who were followed at least 60 days, did not have a pericardial disease, or congenital heart disease were included. Excluded patients included those having a congenital disease with and without valve stenosis (8 patients), followed for <60 days (32 patients) and constrictive pericarditis (3 patients). Consequently, 820 patients were included in the study and were followed for a time period of 60 days to a maximum time period of 2113 days (through October 1, 2016) with a median of 1481 days.
A review of outpatient records was performed to determine the age, sex, and prevalence of the coronary disease, previous myocardial infarction, diabetes, hypertension, and hyperlipidemia. Body surface area, blood pressure, and heart rate at the time of the echocardiogram were recorded. Medications were reviewed at the time of 1st and subsequent visits to determine the use of angiotensin-converting enzyme inhibitors/ angiotensin receptor blockers, beta-blockers, aldosterone antagonists, nitrates, hydralazine, diuretics, calcium channel blockers, digoxin and statins. The coronary disease was defined as having evidence of >50% lesion in any coronary artery by coronary angiography, abnormal myocardial perfusion scan, ECG criteria evidence of an old myocardial infarction [8] or evidence of myocardial infarction using the universal definition [9]. Diabetes was defined as fasting glucose >126 mg%, postprandial blood glucose >200 mg%, hemoglobin A1C >6.5%, or taking anti-diabetic medications. Hypertension was defined as blood pressure >140/90 or taking anti-hypertensive medications. Hyperlipidemia was defined as total cholesterol >200 mg%, triglycerides>150 mg%, or taking lipid-lowering medications. Laboratory data was reviewed and the average brain natriuretic peptide (BNP), hemoglobin, creatinine values, percentage of patients with stage 3 chronic kidney disease as glomerular filtration rate <60 ml/min/1.73 m2 (CKD-EPI study equation [10], and troponin I were recorded. The prevalence of atrial fibrillation was determined by examining the EKG of each patient at each visit or hospitalization. The presence of more than mild aortic valve stenosis, aortic valve regurgitation, mitral, or tricuspid regurgitation was observed from medical records recording clinical assessments of patients. Evidence of verifiable HF was determined using the Framingham HF criteria requiring either 2 major criteria or 1 major and 2 minor criteria (CHF1 score>4) [11] or BNP>100 pg/ml.
Inpatient and outpatient records were examined to determine if patients were readmitted for HF after the initial contact with the health care system by assessing each admission to determine if HF was the reason for admission as opposed to chronic pulmonary disease, pneumonia, or other diagnoses. ACM was determined from medical records, conversation with family, and examining obituaries and/or the national death index. Time from being 1st seen to HF or ACM was determined. The type of specialty caring for the patient was recorded: family medicine, internal medicine, or cardiology. If cardiology was involved in the care of the patient, the patient was assigned to cardiology even if family or internal medicine were seeing the patients.
Patients were assigned to HF phenotypic groups based on LV ejection fraction determined by Doppler echocardiography. LV ejection fraction >50% was HFpEF (n=269). LV ejection fraction <50% was termed HFrEF (HFrEF, n=364). HFrEF was subdivided into HF with mid-range ejection fraction if the ejection fraction was 40-49% and HF with low ejection fraction if the ejection fraction was <40%. Patients without echocardiography performed or other modalities for determination of LV ejection fraction were termed HFunEF (n=187).
2.1. Echocardiography
All images were digitally obtained from 3-5 (5 if atrial fibrillation) consecutive cardiac cycles using a Phillips iE 33 (Cleveland, Ohio) with a 2.5 mHz broadband transducer (X5) at end-expiration. Doppler echocardiography was obtained from multiple ultrasonic windows to provide views of all cardiac chambers and valves. Transmitral pulsed Doppler was obtained from 1-2 x 1-2 mm sample volume placed at the tips of the mitral leaflets. Using a 1-2 x 1-2 mm sample volume, LV outflow tract pulsed Doppler was obtained from the apical 3 or 5 chambers approximately 3-5 mm from the aortic valve annulus. Spectral tissue Doppler was obtained from the septal and lateral mitral annuli using a 3x3 mm pulsed Doppler sample volume. All measurements are the average of 3-5 (5 if atrial fibrillation) consecutive cardiac cycles at end-expiration and were performed according to the recommendation of the American Society of Echocardiography guidelines [12]. We calculated LV end-diastolic and end-systolic volumes (using Simpson’s rule), LV mass, and left atrial volumes and indexed these measures to body surface area. The ejection fraction was calculated in the usual manner. End diastolic basal right ventricular basal dimension and tricuspid annular plane systolic excursion were measured as described by the American Society of Echocardiography guidelines [12]. From the LV outflow tract pulsed Doppler recordings, we determined the peak velocity and velocity-time integral. The LV outflow tract area was calculated as the product of 0.785*LV outflow tract diameter2, assuming circular geometry. The stroke volume index was calculated as the product of the velocity-time integral and LV outflow tract area and indexed to body surface area. From transmitral Doppler, E, A, E/A were obtained based on the American Society of Echocardiography guidelines [13]. Spectral Doppler of the septal and lateral mitral annulus e’ was analyzed and averaged. The E/e’ was calculated [13]. Tricuspid regurgitation velocity was obtained from a continuous wave Doppler transducer using multiple windows. Using the modified Bernoulli equation, right ventricular systolic pressure was estimated with the addition of an estimate of right atrial pressure based on respiratory variations of the inferior vena cava dimension using the American Society of Echocardiography recommendations [13].
2.2. Statistics
Data were expressed as mean+standard deviation for continuous normally distributed data as determined by the Kolmogorov-Smirnov test. For data that were not normally distributed, median and interquartile ranges were computed. Categorical data was expressed as a percentage of the group having that attribute. Differences between the groups were determined using 1-way analysis of variance or 1-way analysis of variance on ranks. If the F value was <0.05, the differences between individual groups were determined by Dunn’s test. Differences in percentages among groups were determined using chi-square. If p<0.05, then a multi-comparison technique was utilized to determine where the significant differences existed. (COMPROP-SAS, Cary, NC). P values <0.05 were considered significant. Multiple logistic regression was performed to determine the independent correlates of ACM and readmission using univariate statistics with p<0.10 for each phenotype. Cox proportional analysis was utilized to assess the relation of HF phenotype to ACM and readmission for HF taking into account the variables derived above. Adjusted Kaplan-Meier curves were subsequently constructed to determine HR’s for ACM and readmission for HF. Landmark analysis was performed on all patients already admitted once to determine HR’s for ACM and re-admission a 2nd time for HF. Log-rank testing was performed to determine significance. Statistics were calculated with Sigma Stat 4 (SYSTAT, San Jose, CA) and SAS (Cary, NC).
3. RESULTS
Baseline patient characteristics are summarized in Table 1. The prevalence of HFunEF, HFpEF, and HFrEF was 22.8%, 32.8%, and 44.4%, respectively. The percentage of patients with HFunEF followed by family medicine was 70.2% (p<0.001 vs Cardiology), 27.8% by internal medicine (p<0.001 vs family medicine and p=0.041 vs cardiology), and 2.0% by cardiology. For the entire database, the percentage of patient care was provided by cardiology (14.6%), internal medicine (26.8%), and family medicine (58.6%). As compared to patients with HFunEF, HFpEF patients were younger, had a greater prevalence of the coronary disease, and hyperlipidemia. As compared to HFunEF patients, patients with HFrEF had a greater prevalence of coronary disease, myocardial infarction, hyperlipidemia, beta-blocker use, aortic regurgitation, mitral and tricuspid regurgitation, lower diastolic and mean blood pressures, greater BNP levels, and lower hemoglobin.
| - | HFunEF (n=187) | HFpEF (n=269) | HFrEF (n=364) |
|---|---|---|---|
| Age (years) | 73+16 | 66+17*** | 75+13^^ |
| Sex (M/F) | 101/130 | 165/104 | 187/177 |
| Coronary Disease (%) | 47.2 | 60.0* | 66.1* |
| Myocardial Infarction (%) | 20.0 | 26.0 | 41.0**^ |
| Diabetes Mellitus (%) | 54.1 | 59.9 | 46.1^ |
| Hypertension (%) | 82.3 | 91.0 | 88.5 |
| Hyperlipidemia (%) | 60.6 | 75.8* | 77.1* |
| Aortic Stenosis (%) | 5.2 | 12.6 | 15.2 |
| Aortic Regurgitation (%) | 2.6 | 3.1 | 13.0*^ |
| Mitral regurgitation (%) | 11.8 | 19.3 | 32.4***^ |
| Tricuspid Regurgitation (%) | 16.1 | 24.5 | 31.6** |
| ACEI/ARB (%) | 62.2 | 68.0 | 65.4 |
| Atrial fibrillation (%) | 22.1 | 21.6 | 24.9 |
| Beta Blockers (%) | 59.4 | 59.2 | 72*^ |
| Diuretics (%) | 78.2 | 72.9 | 80.7 |
| Aldosterone blocker (%) | 9.4 | 18.6 | 12.2 |
| Nitrates (%) | 6.2 | 7.4 | 9.5 |
| Hydralazine (%) | 3.5 | 6.7 | 6.4 |
| Digoxin (%) | 7.4 | 7.4 | 9.5 |
| Calcium Channel Blockers (%) | 17.6 | 19.0 | 24.6 |
| Systolic BP (mm Hg) | 132.5+26.1 | 131.8+23.2 | 128.2+23.1 |
| Diastolic BP (mm Hg) | 73.4+12.2 | 71.1+12.5** | 71.3+12.1* |
| Mean Arterial BP (mm Hg) | 93.1+14.5 | 93.5+15.0 | 90.1+14.2*^ |
| Heart Rate (beats/min) | 79.1+16.4 | 78.6+15.5 | 76.4+14.2 |
| BSA (m2) | 1.89+0.29 | 2.23+0.26*** | 1.79+0.17***^^ |
| BNP (pg/ml) | 677+910 | 477+739 | 818+1067* |
| Hemoglobin (g/dl) | 12.7+2.2 | 12.7+2.2 | 12.0+1.9**^^^ |
| Sodium (mmol/l) | 138.2+4.6 | 137.4+3.2 | 137.3+3.3 |
| Blood Urea Nitrogen (mg/dl) | 24.0+14.1 | 23.0+13.5 | 24.1+13.7 |
| Creatinine (mg/dl) | 1.30+1.00 | 1.28+0.78 | 1.38+0.98 |
| Troponin I (ng/ml) | 0.188+1.244 | 0.724+4.512 | 0.354+2.112 |
Table 2 summarizes differences between patients with HF and mid-range ejection fraction (40-49%) and patients with HF and low ejection fractions (<40%). Patients with ejection fraction<40% comprised 36.5% of the HFrEF group. As compared to HF with mid-range ejection fraction, patients with HF and ejection fraction <40% had a lower prevalence of coronary disease but a more left anterior descending disease with apical akinesis, less hypertension and hyperlipidemia, and lower creatinine.
| - | HFmrEF (n=231) | HFlowEF (n=133) |
|---|---|---|
| Age (years) | 74+12 | 76+14 |
| Sex (M/F) | 101/130 | 65/68 |
| Coronary disease (%) | 64.9 | 54.1** |
| Myocardial Infarction (%) | 32.9 | 24.0 |
| Left anterior descending disease (%) | 39.2 | 59.1* |
| Apical akinesis (%) | 32.3 | 47.7* |
| Diabetes Mellitus (%) | 46.8 | 44.4 |
| Hypertension (%) | 92.2 | 85.0* |
| Hyperlipidemia (%) | 81.0 | 69.9* |
| ACEI/ARB (%) | 64.5 | 63.2 |
| Atrial fibrillation (%) | 19.5 | 26.3 |
| Beta blockers (%) | 72.3 | 71.4 |
| Diuretics (%) | 79.4 | 83.1 |
| Aldosterone blocker (%) | 10.0 | 14.3 |
| Nitrates (%) | 9.0 | 9.8 |
| Hydralazine (%) | 9.8 | 6.8 |
| Digoxin (%) | 10.8 | 15.0 |
| Calcium channel blockers (%) | 23.8 | 25.6 |
| Systolic Blood Pressure (mm Hg) | 127.3+23.0 | 128.3+23.7 |
| Diastolic Blood Pressure (mm Hg) | 71.3+11.9 | 71.3+12.1 |
| Mean Arterial Pressure (mm Hg) | 90.0+14.1 | 90.3+14.1 |
| Heart Rate (beats/min) | 75.9+14.2 | 76.5+13.8 |
| Body surface area (m2) | 1.88+0.09 | 1.62+0.14*** |
| BNP (pg/l) | 803+1069 | 825+1080 |
| Hemoglobin (g/dl) | 12.0+1.9 | 11.9+2.0 |
| Creatinine (mg/dl) | 1.47+1.13 | 1.20+0.73* |
| LVEDVI (ml/m2) | 58.0+25.0 | 55.4+19.2 |
| LVESVI (ml/m2) | 32.9+22.5 | 34.6+16.3 |
| Ejection Fraction (%) | 46.5+2.3 | 37.4+4.5*** |
| Left atrial volume index (ml/m2) | 35.4+17.3 | 37.0+14.3 |
| E (cm/s) | 101.8+35.2 | 110.6+37.9* |
| E/e’ | 14.3+6.5 | 15.4+7.3 |
| Stroke volume index (ml/m2) | 37.2+14.5 | 41.3+17.8 |
| Tricuspid Regurgitation velocity (m/s) | 2.86+0.6 | 2.93+0.6 |
| CHF1 score>4 or BNP>100 pg/ml | 60.3 | 56.3 |
| 1st Readmission rate (%) | 44.6 | 45.1 |
| 2nd Readmission rate (%) | 23.3 | 23.3 |
| 2nd Readmission rate- previously readmitted (%) | 51.5 | 53.1 |
| ACM rate (%) | 32.4 | 38.9 |
| - | HFpEF (n=269) | HFrEF (n=364) |
|---|---|---|
| LVEDVI (ml/m2) | 51.9+20.5 | 57.1+23.2** |
| LVESVI (ml/m2) | 29.3+19.4 | 31.3+20.2* |
| Ejection Fraction (%) | 55.2+4.0 | 41.3+20.6*** |
| LV Mass Index (g/m2) | 98.3+30.8 | 100.1+31.0 |
| Left atrial volume index (ml/m2) | 32.7+14.3 | 38.7+18.6** |
| E (cm/s) | 100.3+33.8 | 74.6+58.9* |
| E/A | 1.26+0.65 | 1.33+0.74 |
| Deceleration time (msec) | 223.1+73.2 | 216.9+74.7 |
| E/e’ | 13.3+6.0 | 14.8+6.9 |
| Stroke volume index (ml/m2) | 35.0+14.1 | 36.7+15.9 |
| TR velocity (m/s) | 2.78+0.59 | 2.88+0.60 |
| RAP (mm Hg) | 7.2+4.4 | 7.7+4.7 |
| TAPSE (mm) | 13.9+6.1 | 14.0+5.3 |
| RV base (cm) | 3.9+0.9 | 3.8+0.8 |
| - | HFunEF (n=187) | HFpEF (n=269) | HFrEF (n=364) |
|---|---|---|---|
| Verifiable HF: CHF1 score>4 or BNP>100 pg/ml | 30.9 | 57.9** | 58.9** |
| 1st HF Readmission rate (%) | 16.0 | 40.1*** | 45.1*** |
| 1st HF readmission (days) | 1864+1935 | 1372+1190*** | 1431+1699** |
| 2nd HF readmission rate based on previous readmission rate (%) | 25.0 | 42.6* | 53.1** |
| 2nd HF readmission based on previous readmission (days) | 156+449 | 316+540*** | 285+521** |
| ACM (%) | 43.4 | 26.7* | 34.2 |
| ACM (Days) | 1668+1046 | 1668+1045 | 1674+1668 |
Table 3 summarizes the Echo-Doppler parameters for HFpEF and HFrEF. Patients with HFunEF did not have echocardiograms performed. Patients with HFrEF had greater LV and left atrial volume indexes and a lower LV ejection fraction.
Table 4 summarizes HF readmission and ACM for all 3 HF phenotypes. As compared to HFunEF, patients with HFpEF were greater in number with verifiable HF, higher HF readmission rate in a fewer number of days, a higher 2nd HF readmission rate for patients admitted once for HF in a fewer number of days, and a lower ACM. As compared to HFunEF, patients with HFrEF demonstrated a greater percentage of patients with verifiable HF, higher HF readmission rate in a fewer number of days, and a higher 2nd HF readmission rate for patients admitted once for HF in a fewer number of days.
Table 5 summarizes the multiple logistic regression analysis for HF readmission, 2nd HF readmission for patients already readmitted once, and ACM for all phenotypes. For HFunEF, diabetes and ACM were independently related to HF readmission. Age, diabetes, and HF readmission were independently related to ACM. For HFpEF, ACM was independently related to HF readmission. For 2nd HF readmission for patients previously readmitted, BNP>100 pg/ml was independently related. For ACM, HF readmission, age, mitral regurgitation, and hemoglobin were independently related. For HFrEF, BNP>100 pg/ml and beta-blocker use (inverse relation) were independently related to HF readmission. BNP>100 pg/ml was independently related to the 2nd HF readmission for patients previously readmitted. BNP >100 pg/ml, hemoglobin, and age were independently related to ACM.
| - | HF with Unknown Ejection Fraction | HFpEF | HFrEF |
|---|---|---|---|
| HF Readmission |
Diabetes OR:3.29(1.38-8.25),p=0.011 |
ACM OR:2.46(1.23-4.91),p=0.011 |
BNP>100pg/ml OR:4.78(2.99-7.57),p<0.001 |
| - |
ACM OR:2.40(1.01-3.78), p=0.049 |
- |
Beta blocker use OR:0.47(0.25-0.88),p=0.019 |
| 2nd HF Readmission for patients previously readmitted | none |
BNP>100 pg/ml OR:1.81(1.03-2.89),p=0.032 |
BNP>100 pg/ml OR:1.78(1.01-3.52),p=0.041 |
| ACM |
Age OR:1.06(1.02-1.76),p=0.007 |
HF Readmission OR:2.98(1.62-5.51),p<0.001 |
BNP>100 pg/ml OR:2.27(1.38-3.73),p<0.001 |
| - |
Diabetes OR:2.44(1.19-4.98),p=0.015 |
Age OR:1.07(1.03-1.92),p<0.001 |
Hemoglobin OR:0.80(0.71-0.91),p=0.006 |
| - |
HF Readmission OR:1.48(1.01-1.67),p=0.046 |
Mitral regurgitation OR:2.52(1.09-5.82),p=0.031 |
Age OR:1.03(1.01-1.05),p=0.017 |
| - | - |
Hemoglobin OR:0.88(0.75-0.99),p=0.049 |
- |
Fig. (1A) depicts the adjusted Kaplan-Meier curves for ACM for each phenotype over the length of follow-up and indicates that HFunEF had significantly greater ACM than HFpEF (HR=1.60, p=0.004), and HFrEF had significantly greater ACM than HFpEF (HR=1.84, p=0.043). When HFrEF is subdivided (Fig. 1B), there was no significant difference in ACM between subgroups with LV ejection fraction <40% and LV with mid-range ejection fraction=40-49%.
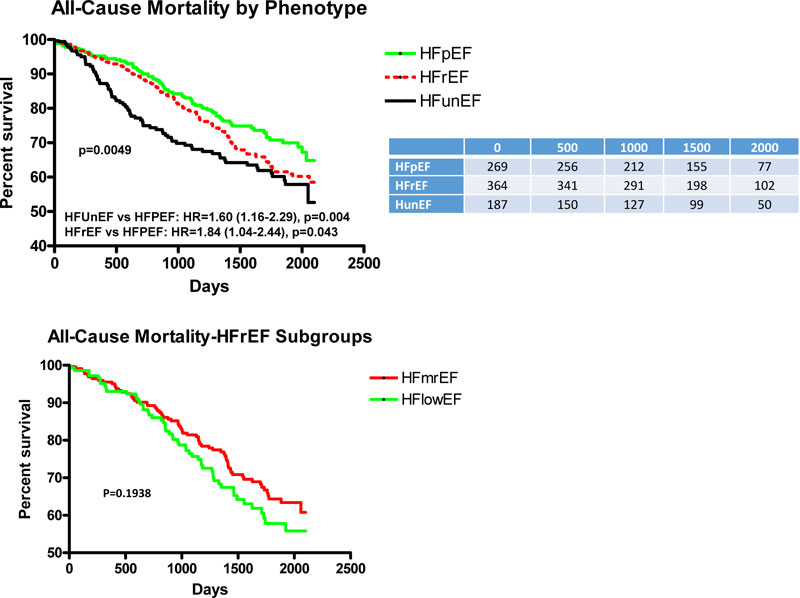
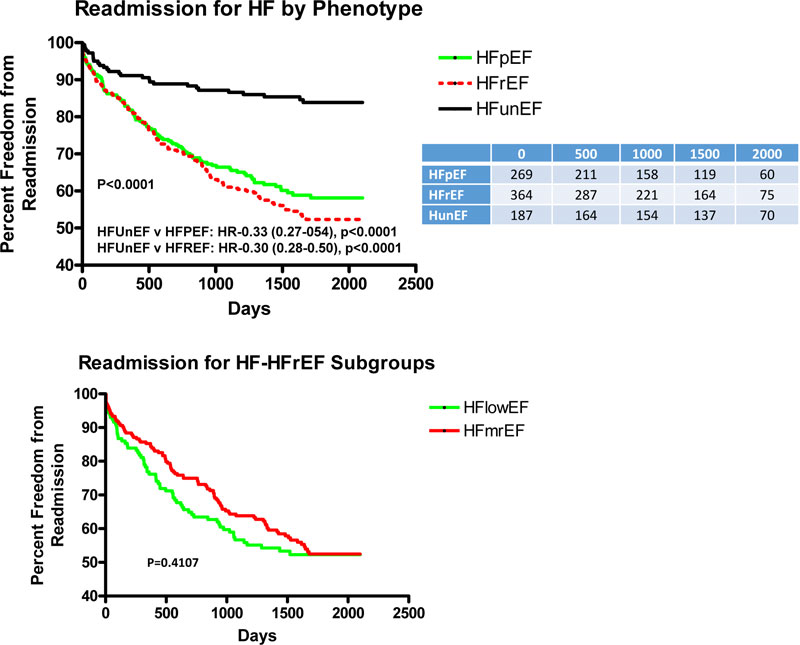
Fig. (2A) depicts the adjusted Kaplan-Meier curves for HF readmission for all HF phenotypes over the length of follow-up. There was a significantly lower rate of readmission for HFunEF as compared to both HFpEF and HFrEF with HR’s of 0.28 (p<0.0001) and 0.30 (p<0.0001), respectively, with similar HF readmission between the 2 HFrEF subgroups (Fig. 2B).
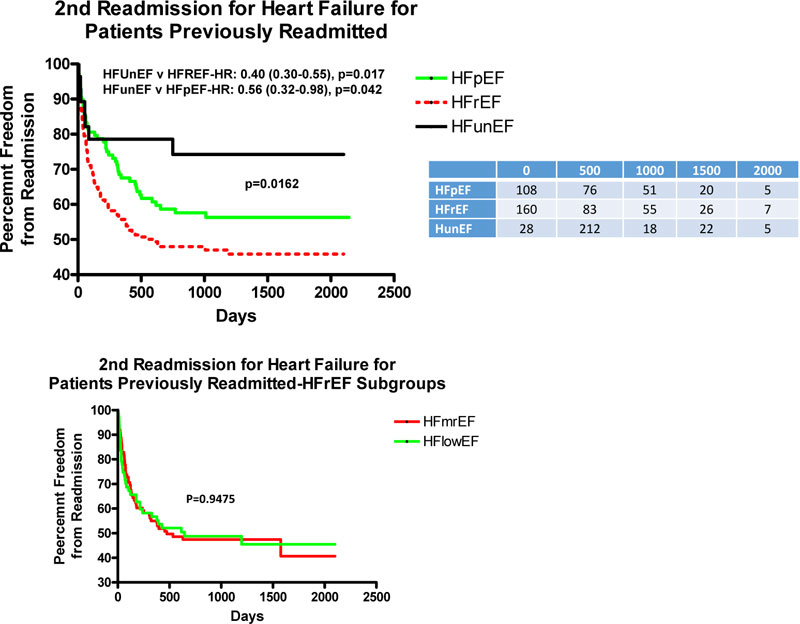
Fig. (3A) depicts the adjusted rate of 2nd HF readmission for patients previously readmitted for HF. There was a lower rate of 2nd HF readmission over the length of follow-up for patients with HFunEF as compared to HFrEF and HFpEF with HR of 0.40 and 0.56, respectively, but the curves are superimposable for the 2 subgroups of HFrEF (Fig. 3B).
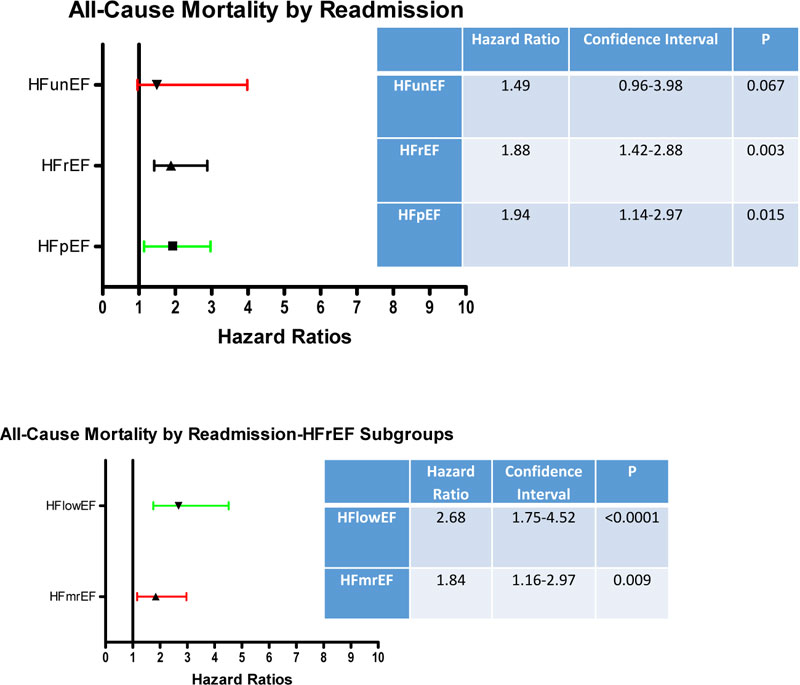
Fig. (4A) demonstrates increased ACM in patients with HFpEF and HFrEF who have been previously readmitted. There was only a trend for patients with HFunEF with increased ACM with prior HF readmission. When HFrEF was divided into subgroups, increased HRs were noted with both subgroups (Fig. 4B)
4. DISCUSSION
In this study, we demonstrated that in a rural community cohort, 22.8% of patients with a diagnosis of HF did not have an ejection fraction calculation and 70% were cared for by family medicine. Second, patients with HFunEF had a lower prevalence of coronary disease, hyperlipidemia and lower diastolic blood pressure. Third, adjusted ACM was increased as compared to HFpEF and HF readmission was reduced as compared to the other HF phenotypes possibly related to the increased ACM. Furthermore, the 2nd readmission in patients previously readmitted was also reduced as compared to the other HF phenotypes. Fourth, ACM based on prior readmission only showed a positive trend as compared to significant increases in HFpEF and HFrEF. Finally, although differences were noted in subdividing HFrEF into subgroups with HF with LV ejection fraction<40% or HF with LV ejection fraction between 40-49%, ACM and HF readmission were similar.
4.1. Previous Literature
HF is a chronic problem associated with high morbidity and mortality with higher prevalence and incidence in the elderly population due to increased survival from myocardial infarction and increased prevalence of co-morbidities with aging. Diagnosis can be easily missed due to non-specific symptoms that simulate respiratory conditions and lack of access to or use of echocardiography. Diagnosis of HF can be further substantiated and phenotyped with echocardiographic assessment characterized by either HFrEF or HFpEF. According to the European Society of Cardiology, HF is clinically defined as a syndrome in patients with typical symptoms (shortness of breath, fatigue and ankle swelling) and signs (elevated jugular venous pressure, pulmonary crackles, and displaced heartbeat) resulting from an abnormality in cardiac structure and function [14]. The diagnosis of HFrEF requires 3 criteria: symptoms, signs as stated above, and a reduced LV ejection fraction. The diagnosis of HFpEF needs 4 criteria: typical symptoms, signs, LV ejection fraction >50%, and structural heart disease including LV hypertrophy, left atrial enlargement and/or diastolic dysfunction. However, there are patients who are not “phenotyped” in administrative or even patient-level databases because an assessment of LV ejection fraction was not performed. This study addresses the characteristics and some of their outcomes from a single rural database in a community medical school without a specific HF service.
Explanations as to why patients without an ejection fraction determination should demonstrate increased ACM are at best speculative. Differences in clinical characteristics include a lower prevalence of coronary disease, hyperlipidemia, mitral and aortic regurgitation as compared to HFrEF. These differences, except for coronary disease, are small and unrelated to ACM by multiple logistic regression. The lower prevalence would not have an obvious mechanistic reason to increase ACM. Additionally, HF medication (angiotensin-converting enzyme inhibitors/ angiotensin receptor blockers, beta-blockers, and aldosterone antagonists) that is used to promote survival, was limited in this group and similar to the other groups. Of note, readmission for HF was lower and consequently, attention to HF care may have been reduced in this group. We were unable to characterize the cause of ACM, whether it is non-cardiac or cardiac and the prevalence of sudden death.
Previous studies have reported an increased prevalence of unrecognized HF in elder populations referred to a cardiologist or to the hospital for echocardiography to confirm the diagnosis. Riet et al. [15], studied 585 patients >65 years old presenting to the primary care physician with dyspnea on exertion in the last 12 months but no confirmatory echocardiogram to determine the prevalence of HF in the elderly population. These patients underwent history examination, physical exam, electrocardiography and serum N-terminal pro-B-type natriuretic peptide testing with echocardiography performed only in patients with abnormal electrocardiography or serum N-terminal pro B-type natriuretic peptide >15 pmol/ml. Using European Society of Cardiology Guidelines, 92 patients had HF, 17/585 (2.9%) had HFrEF and 70/585 (12%) had HEpEF, while 5 (0.9%) had isolated right-sided HF. They also noted that HF was confused with chronic obstructive pulmonary disease and other respiratory problems in the primary care setting, which could lead to delay in diagnosis resulting in increased HF hospitalization, morbidity and mortality. Winter et al. [16], demonstrated the prevalence of unknown HF and LV dysfunction in patients >60 years of age with type 2 diabetes mellitus. Patients were diagnosed with HF on the basis of the European Society of Cardiology Guidelines. Of the 581 patients studied; 161 were diagnosed with HF: 28 (4.8%) had HFrEF and 133 (22.9%) had HFpEF. There was increased prevalence with age and a female sex preponderance. Barents et al. [17], reported the prevalence, misdiagnosis of chronic HF and the role of natriuretic peptides in 150 nursing home residents using the New York Heart Association classification and natriuretic peptides. HF diagnosis was confirmed in 24 residents, out of which, 15 had de-novo HF. Another important feature is that 13 out of 22, who were thought to have HF before screening, were rejected after the screening. Rutten et al. [18], reported the prevalence of HF in an elderly population with stable chronic obstructive pulmonary disease in whom patients had a detailed history, physical exam, chest radiography, ECG, echocardiography and pulmonary function testing. The diagnosis of HF was based on the European Society of Cardiology guidelines. Unrecognized HF was noted in 83 of 405 patients. Of the 83 patients, 42 had HFrEF and 41 had HFpEF. Most males who were younger had HFrEF, while HFpEF was common in elder women. Finally, Mosterd et al., included 5450 participants aged 55-95 years in the Rotterdam study to determine the prevalence of HF and symptomatic or asymptomatic LV systolic dysfunction. The overall prevalence of HF was 3.9%, with no difference between men and women, with an increased prevalence with age. The prevalence of LV systolic dysfunction was 3.7% and 2.5 times more common in men and most were asymptomatic. Most of the previous literature commented on the characteristics of populations screened for LV dysfunction or HF in variably symptomatic populations. There is little data characterizing patients with HFunEFand their outcomes, most likely due to national programs and core hospital measures advocating and requiring an assessment of LV ejection fraction. Patient data and outcomes are often not evaluated in this subset of patients.
4.2. Limitations
As this was a retrospective evaluation of a community-based outpatient practice, it is limited by numbers and population diversity and the inherent nature of a retrospective study. Second, data abstraction was based on a chart review of both inpatient and outpatient recorded symptoms and medications. ACM was based on chart review, national death index, and obituary review and may not have reflected all mortality. Cardiovascular mortality could not be discerned nor medication compliance. Furthermore, optimal dosing was not evaluated in this study with regard to outcomes because of changing dosages in many patients for unclear reasons despite chart abstraction. Third, readmissions for HF may have occurred outside the East Tennessee area. The additional hospital system’s records in the area were available and reviewed for admissions as patients may use both systems. Fourth, laboratory values were not available in all patients for BNP (7% missing), with non-significant differences among HF phenotypes ranging from 5-10%. Fifth, as there is inherent variability in the measurement of ejection fraction, this may lead to assigning patients to the wrong phenotype. As this was a retrospective study, there may be additional unknown differences among HF phenotype groups that were not accounted for.
4.3. Clinical Implications
Despite the requirement for hospital HF core measures for ejection fraction determination for all inpatients being observed in >90% cases, there are still patients with HF diagnosis who have do not have an assessment of ejection fraction. Furthermore, there appears to be an underutilization of guideline-based medications. The majority of such patients were seen by non-cardiovascular providers in this study, but is likely true in many populations. Unfortunately, these patients are readmitted less but have greater ACM. Demographic variables and laboratory variables, including brain natriuretic peptide, show little difference. Although this study portrays them as a separate phenotype, they clearly are not. More importantly, the lack of structural information afforded by echocardiography represents a lost opportunity and may have a role in their outcomes. It is also not inconceivable that non-cardiac causes may predominate their causes of death. The degree of detail and attention paid to ambulatory HF programs will need to be focused on all providers to afford patients’ optimal care and outcomes.
CONCLUSION
In patients with HFunEF, ACM is increased and readmission for HF is reduced as compared to other phenotypes. The lack of structural information afforded by echocardiography may impact the care provided especially by non-cardiovascular providers in a rural community-based setting.
LIST OF ABBREVIATIONS
| ACM | = All-cause mortality |
| BNP | = Brain natriuretic peptide |
| HFunEF | = HF with unknown ejection fraction |
| HF | = Heart failure |
| HR | = Hazard ration |
| HFrEF | = HF with reduced ejection fraction |
| HFpEF | = HF with preserved ejection fraction |
| LV | = Left ventricular |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This stude has been approved by East Tennessee State University, USA IRB-0515.18sw.
HUMAN AND ANIMAL RIGHTS
Not applicable.
CONSENT FOR PUBLICATION
Informed consent was obtained from all participants.
AVAILABILITY OF DATA AND MATERIALS
The data in the manuscript can not be shared because the data despite being de-identified is obtained from a small rural area with a limited population using the University medical practice. The possibility of patient anonymity can not be guaranteed.
FUNDING
This study has been funded by American Heart Association.
CONFLICTS OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
We would like to acknowledge the support from the American Heart Association from the summer student program in support of Danielle Kelvas between her 1st and 2nd year of medical school.


