Pharmacokinetic Considerations for Digoxin in Older People
Abstract
Objective:
This review aims to arm readers with a deep understanding of pharmacokinetics of digoxin.
Data Sources:
Pharmacology and pharmacokinetic references texts, and peer reviewed medical journal manuscripts indexed on Medline included based on currency, accuracy and appropriateness.
Results:
Results: Physiologic changes and disease associated with aging have an impact on pharmacokinetics and pharmacodynamics of medications. Altered drug response and increased adverse reactions are common amongst the elderly. The narrow therapeutic index of digoxin and pharmacokinetic changes associated with aging increases the risk of toxicity. In the aging population, a number of factors combine to increase the risk, severity and likelihood of hospitalisation or death due to adverse drug effects:
- changes to absorption, distribution, metabolism and excretion,
- increased susceptibility to drug sensitivity,
- co-existing pathology,
- polypharmacy.
Conclusion:
A thorough understanding of digoxin pharmacokinetics in the older person is essential for improved therapeutic outcomes, improved compliance, reduced morbidity and improved quality of life.
INTRODUCTION
Pharmacokinetics examines the absorption, distribution, metabolism and excretion (ADME) of drugs, and the associated toxic or therapeutic responses [1-4]. Pharmacokinetics includes applications in bioavailability, variations due to physiological or pathological conditions, disease related dose adjustment, drug interactions, and customisation of drug dosage regimes [1-4]. An important division of pharmacology closely related to pharmacokinetics is the study of factors affecting bioavailability to optimise therapeutic activity of drugs, known as biopharmaceutics [1, 2].
The underlying principle of pharmacokinetics and the focus of this discussion is consistent with the philosophy of Paracelsus (medieval alchemist) who suggested that “only the dose makes a thing not a poison” [1]. Within a window, a specific drug will offer therapeutic benefit and outside that window there will either be no therapeutic benefit or toxicity. The narrow therapeutic range of digoxin means that small variations in blood concentration may easily result in toxic or sub therapeutic concentrations. To maintain concentrations within the therapeutic range requires consistent bioavailability and careful management of factors that may influence bioavailability. Thus the aging body presents variations to physiological and pathological status that can have a profound influence on bioavailability, pharmacological response, drug sensitivity and drug interactions. The changes associated with the elderly demand more astute medication management and monitoring.
DRUG RESPONSE AND AGING
Physiologic changes and disease occur with aging and can affect drug pharmacokinetics in older persons [5, 6]. Older people can not only have altered function but they can also have altered responses to the drugs themselves relating to mechanical responses, receptor mechanisms, homeostatic changes and CNS function [7,8]. The high prevalence of disease in the elderly also results in a higher use of medications and the incidence of adverse drug effects correlates with age [9]. As many as 20% of hospitalisations in the elderly are due to adverse effects of medications and 18% of hospital deaths in the elderly are associated with adverse effects of medications [9]. Perhaps the most important consideration for drug use and response in the elderly is that there is greater heterogeneity in older populations than younger people which means not only that there is significant variation is disease states but also significant variations in responses to medications [10]. No doubt the under representation of the elderly in pharmaceutical clinical trials contributes to adverse effects in this cohort [9].
With aging comes:
- Altered absorption (eg. slower gut or transdermal absorption).
- Changed bioavailability (eg. increased for highly extracted drugs).
- Changed biodistribution (eg. more extensive for lipid soluble drugs and less extensive in water soluble drugs).
- Altered metabolism (eg. cytochrome based metabolism in the liver).
- Altered elimination (eg. slower renal excretion) [5, 6, 11].
Irrespective of age related changes to absorption, distribution, metabolism and excretion, older people also demonstrate an increased sensitivity to many drugs due to comorbidity and polypharmacy [7]. The elderly are at higher risk of an adverse drug effect, have increased severity of effects, are less likely to report adverse effects and are more likely to be hospitalised or die due to adverse drug effects [9].
Pharmacokinetic changes due to age related physiological variations demand attention toward dose requirements in older people [5, 6]. Nonetheless, it is often difficult to differentiate pharmacokinetic changes resulting from aging from coexisting environmental influences, disease and other medications [12]. Consequently, drug therapy is difficult to predict in an older person so medication should be started at a low dose and increased this slowly with close monitoring of benefits and adverse effects [12, 13].
Absorption
With increasing age the amount of saliva produced is often reduced and this can reduce the rate of drug absorption by influencing the gastric pH [2, 8]. Furthermore, older people have reduced gastric acid secretion and reduced acidity (increased pH) which can delay dissolution of oral medications [2, 3, 8, 9]. This is exacerbated by delayed gastric empty due to reduced peristaltic force that reduces the mechanical influences on medication mixing with gastric juices [3, 8, 9]. The surface area for drug absorption is also decreased in aging due to intestinal atrophy which, combined with reduced concentration gradient due to the poorer blood flow, inhibit passive diffusion of drugs into the blood stream further delaying absorption rate [8]. While this does not change the total absorption itself, it does alter the time to maximum drug effect [3, 8]. Generally speaking, most orally administered drugs (like digoxin) are not required to be altered on the basis of age related changes in gastrointestinal function [8]. A number of studies have shown factors contributing to absorption are generally not physiological changes but rather to various disease states [11].
Distribution
Drug distribution in the older person can vary due to changes in body composition and critical organ perfusion (due to reduced cardiac output and increased peripheral vascular resistance) [3, 8]. The latter will also affect metabolism and elimination with decreased liver and kidney perfusion. The reduction in lean body mass in older people (as much as 19%) will cause an elevation in drug concentrations in muscles for drugs distributed in that manner (eg. digoxin) [8,14]. The older body also sees an increase of up to 20-40% in the relative fat composition (adipose tissue) independently of obesity and, despite the increase in fat, older people have a marked decrease in water content of the body of 10-15% [8, 9, 14]. The effects of these changes on drug distribution will largely depend on the lipid or water solubility nature of the drug [8] and its metabolites. Water soluble drugs (like digoxin) will show a small volume of distribution (V), increased plasma concentrations with reduced total body water and, thus, accommodation needs to be made in reducing loading doses [8,11,14]. Conversely, lipid soluble drugs, will have a larger volume of distribution and, thus, a longer half life [11]. The half life of water soluble drugs does not reduce because of the concomitant reduced renal clearance. From the equation below, it is clear that a reduction in V in older patients with concomitant reduction in renal clearance will cancel one another out while the renal reduction potentiates the effects of increased V on half life. A 10% decrease in blood albumin concentration has the effect of increasing the unbound fraction of the drug by a corresponding 10% [9].
T1/2 = [0.693 x V] / renal clearance
Metabolism
The vast majority of drug metabolism occurs in the liver with minor contributions from the kidney and lung [8]. The capacity of the liver to metabolise drugs decreases with age [6]. The 35-40% reduction in hepatic perfusion in the elderly has a significant impact on drug delivery to the liver and subsequent rate of metabolism [8, 12, 13]. Liver size can be reduced by about 24-35% with age [13]. One major influence is the reduced first pass metabolism of orally absorbed lipid soluble drugs which can significantly increase drug bioavailability [8]. Conversely, prodrugs that require metabolism for activation may show decreased or delayed bioavailability [11]. Digoxin is not strongly influenced by first pass metabolism or hepatic metabolism. While no significant decrease occurs in phase II metabolism, there is a decrease in clearance of drugs metabolised by phase I pathways [9]. Furthermore, the decreased capacity (up to 60%) of the liver to metabolise drugs for elimination can increase plasma concentrations and prolong biological half lives of drugs [8]. It has been recently reported that age related renal dysfunction contributes to decreased liver metabolism of drugs in older people [11]. Finally, the reduction in first pass metabolism and decreased liver capacity to manage toxins contribute to an increase in adverse effects in the elderly.
Elimination
For water soluble drugs, like digoxin, elimination is solely or primarily via renal excretion [8, 9, 11]. In the elderly (compared to young adults), the kidneys can show a 20% reduction in size, 30% reduction in glomeruli function (elimination of drugs relates to glomeruli filtration rate), and as much as 50% reduction in normal renal capacity [8]. This functional decline can be further exacerbated by concomitant renal disease [3, 8]. Renal excretion of drugs declines with age so the elderly should be managed as though they have renal insufficiency [6, 14]. This is particularly important for drugs with a narrow therapeutic index, like digoxin, because it increases the likelihood of serious toxicity [11].
While plasma creatinine levels are a poor indicator of renal function, creatinine clearance rates can be used to accommodate loading and continuing drug doses in the elderly using dose corrections like the Cockcroft and Gault equation [8, 9]. This is essential for drugs that are highly toxic and which have a narrow therapeutic range like digoxin [8] and will be discussed in more detail below. But it is also very important to consider the effects of acute illness in the elderly that can cause a very rapid change in renal function and, for example, see a stable drug regime rapidly degenerate into toxicity following an acute illness (eg. chest infection) [8].
Compliance and Concordance
Compliance is simply defined as the extent to which a patient adheres to the instructions provided for a medication [15]. Concordance is a variation on this concept that accommodates a negotiation of the instructions between prescriber and patient to develop a medication regime that suits the patient and offers therapeutic benefit [10]. Clearly the concept of concordance is aimed at addressing perceived issues in compliance. Perhaps the perception that the elderly are less compliant with medications actually relates more to medication errors than deliberate lack of compliance. Indeed, as few as 18% of the elderly in one study had compliance related problems with their medication while as many as 50% of the elderly make errors in their medications [15].
Using digoxin as an example, compliance can have several significant effects. Firstly, lack of compliance can result in a reduced therapeutic effect which may be difficult to differentiate from disease progression. Secondly, the dose regime may be changed (increased dose) if compliance is not identified as being problematic. Improved compliance after dose adjustment following a period of poor compliance could result in toxicity.
Polypharmacy
Polypharmacy is very important in the elderly because it relates to the relationship between the greater number of medications taken (typically high in the elderly) and the increased risk of drug interaction, decreased compliance and increased medication errors [9,13,15-17]. Indeed, medication errors increase 15 fold with an increase in medications from 1 to 4 [15]. Polypharmacy is defined by five or more prescription medications which has an incidence of 20-40% in the over 70 age group [9]. Older patients are frequently prescribed multiple drugs which make them susceptible to drug-drug interactions [9, 16-19]. Moreover, older people take more drugs with a narrow therapeutic index, increasing the risk of adverse reactions and drug interactions [16]. Unfortunately, they are also less capable of coping with reaction and interactions due to co-morbidity and altered pharmacokinetics [16, 17].
DIGOXIN
Cardiac glycosides come from foxglove (digitalis) and related plants [20, 21] and have been used medicinally (or as a poison) since 1500BC [22]. The digitalis leaf was originally reported (1785) for its role as a diuretic although its influence on cardiac parameters was noted [22, 23]. It was not until the second half of the 19th century that the effects of digitalis glycosides on heart contraction were obvious [23]. It was only approved by the Food and Drug Administration (FDA in USA) for use in heart failure in 1998 [24].
The mechanism of action of cardiac glycosides is generally the same with only minor pharmacokinetic differences between individual agents (eg. digoxin, digitoxin and oubain) [25]. Digoxin remains the most widely used cardiac glycoside (digitalis glycoside) despite the emergence of newer agents because it has few side effects when appropriate dosing is guided by serum concentrations and because it does not increase long term mortality [19, 21, 22, 24,25].
Digoxin is incompletely absorbed (oral dose) and has a substantial fraction cleared by kidneys [4] with 70-85% excreted in urine unchanged [22, 23, 25]. The bioavailability of digoxin varies from 50% to more than 90% of the oral dose [4, 23, 25] although this increases for gelatin capsules to nearly 100% (intravenous is assumed to be 100%) [4, 25].
Digoxin doses are typically 0.01-0.02 mg/kg for loading doses and then 0.125-0.25 mg/day for maintenance [4,24] and plasma concentrations of 1-2ug/L are considered to be the limits of the therapeutic range [4, 23] but this has been revised recently to 0.5-1 ug/L for congestive heart failure [4]. The half life of digoxin is 1.5-2 days (36-44 hours) although this becomes prolonged in renal dysfunction [4, 20-23, 25, 26]. Digoxin has a volume of distribution of about 7.3 L/kg but this decreases in renal disease and hypothyroidism (increases in hyperthyroidism) [4].
Oral digoxin follows a two compartment model (Fig. 1); compartment one (small volume) being plasma and rapidly equilibrating tissues and compartment two (large volume) being the more slowly equilibrating tissues [4]. Concentrations in heart, liver and skeletal muscle tends to be higher than plasma concentrations [25] and 20-30% remains plasma bound [22, 23, 25]. Since cardiac effects are associated with the larger volume, plasma concentrations are not an accurate reflection of pharmacologic effects until there is equilibrium between both compartments (at least 6 hours post oral dose) [4]. Nonetheless, the distribution half life of 35 min, onset of action (oral) of 30-120 min, and time to peak action of 6-8 hours (oral) means that pharmacological effects can be seen well before equilibrium [4]. The ideal time to sample is 7-14 days post commencement of maintenance regime [4].
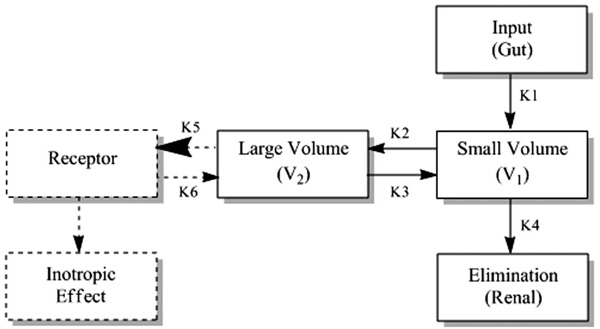
The two compartment model for digoxin.
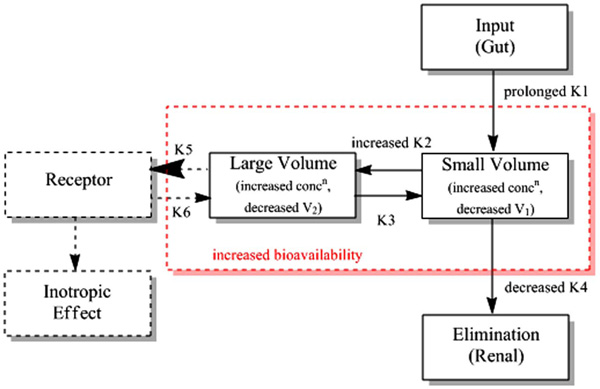
The two compartment model for digoxin in the elderly.
Digoxin has a narrow therapeutic index [1, 23, 25] so small variations in plasma concentration may readily result in toxic or sub therapeutic concentrations. Thus, consistency in bioavailability is important. Moreover, toxic symptoms can be evident with normal digoxin concentrations [25]. Doses should be individualised based on renal function in the first instance and modified based on response and plasma concentrations [25]; this is more important in older people.
Pharmacologic Actions of Digoxin
There are numerous actions of digoxin on the heart; positive inotropism (increased contraction force), negative dromotropism (decreased conduction through the atrioventricular node), and negative chronotropism (decreased heart rate) [20-23, 25, 26]. The inotropic action is due to digoxin inhibiting the sodium-potassium adenosine triphosphatase enzyme (Na+-K+-ATPase) which interferes with the active transport of sodium and potassium across the cell membrane [20-23, 25, 26]. This, in turn, elevates intracellular sodium, increasing calcium availability to produce a more forceful myocardial contraction [20-23, 25, 26]. Chronotropic effects relate to the action of digoxin on automaticity, the ability of the heart to initiate impulses with external stimuli [20, 21, 23, 25, 26]. Therapeutic plasma concentrations of digoxin decrease automaticity while toxic doses can have the opposite effect and increase automaticity [20, 21, 23, 25, 26]. Dromotropic effects relate to the action of digoxin on reducing conduction velocity (directly through atrioventricular node and indirectly through vagal action) and prolonged refractory period [20, 21, 23, 25, 26].
There are also potential benefits of digoxin in the aging patient. Laudisio et al. [27] reported cognitive improvement in patients treated with digoxin. Importantly, cognitive dysfunction is a common co-morbidity in heart failure patients [27, 28]. The cognitive improvement in digoxin therapy relates to the role endogenous cerebral digoxin plays in neuronal sodium dependent transport of calcium [27]. The study involved more than 1100 patients with heart failure and showed cognitive improvement in 25% of digoxin patients and only 16% of non digoxin patients (P < 0.001). In those without heart failure (more than 2400), digoxin improved cognition in 23% compared to just 17% in those untreated (P < 0.001). The investigators corrected for confounders and reported an odds ratio for improving cognition of 1.7 (95% confidence interval of 1.2 to 2.4) for patients with heart failure but only 1.1 (95% CI of 1.0 to 1.3) for those without heart failure. Thus, digoxin therapy appears to selectively improve cognitive performance among older patients with heart failure.
DIGOXIN AND AGING
Despite its narrow therapeutic index and potential fatal toxicity, particularly in the elderly, digoxin is widely used in the management of congestive heart failure and arrhythmia [17, 29, 30]. Not surprisingly given the incidence and prevalence of heart failure in the elderly (80%), digoxin is one of the most frequently prescribed medications in the over 65 year population [10]. Given population aging (relative and absolute) one might expect digoxin use to further increase in older patients [19]. Generally, digoxin is combined with diuretic therapy to reduce the symptoms of heart failure and to increase exercise capability [10]. Unfortunately, digoxin is associated with a significant increase in hospitalisations and mortality amongst the elderly [10]. Digoxin toxicity is common in the elderly [10, 19, 30] and relates to a number of factors [10, 30]:
- Reduction in lean body mass,
- Decreased GFR,
- Decreased muscle mass,
- Diuretic induced potassium loss,
- Drug interactions, and
- Co-morbidity.
Aging results in prolonged elimination half life and decreased volume of distribution for digoxin [3]. A recent examination of more than 1000 nursing home residents in Canada showed that 32% of elderly heart failure patients are treated with digoxin, 80% of those received doses higher than recommended, serum digoxin levels were higher than toxic levels in 30% of patients, and 26% had other medications known to be high risk of digoxin interaction prescribed [29].
The most important age related change is that of deterioration of renal function and this is especially true for digoxin where poorer renal excretion demands lower dosage to avoid toxicity [17]. This is reflected in some early research in normal young and old populations [31]. Using the two compartment model described above, the transfer constant from small to large compartments was increased, elimination was decreased and both of the compartmental volumes of distribution reduced. Fig. (2) provides an overview of the two compartment model for digoxin in older people and the two compartments.
Ewy et al. [32] reported digoxin clearance of 53 ml/min/1.73m2 in older (73-81 years) men and 83 ml/min/1.73m2 in younger men (20-33 years). In a comparison of the pharmacokinetics of digoxin in young (34-61 years) and older (72-91 years) people, Cusack et al. [33] reported a longer biological half life (37 to 70 hours), longer bioavailability (90 to 145 nmol/ml/hr), lower absorption (84% to 76%), decreased volume of distribution (5.3 to 4.1 L/Kg) and slower plasma clearance (106 to 37 ml/min) for the older cohort.
The symptoms of digoxin toxicity in the elderly are protean with the more common features including anorexia, cognitive changes, hazy vision, and arrhythmia [3, 25]. The latter three are not typical of digoxin toxicity in younger populations (anorexia and vomiting) and are consistent with numerous other physiological or pathological process in the elderly (poor specificity) making diagnosis of toxicity difficult [10, 19, 30].
Serum digoxin concentrations are a good marker for toxicity but should be interpreted with heavy weighting on clinical judgement because serum concentrations are not always a good indicator of toxicity [30]. This partly relates to the wide variety of co-morbid conditions and medications that can interfere with the reliability of the serum measurement of digoxin; factors more prevalent in the elderly. With reference to the two compartment model, serum concentrations reflect the small volume while the pharmacologic effects and toxicity are associated with the larger volume. Thus, it is particularly important that equilibrium between the two compartments is reached before serum concentrations are measured. The time to equilibrium can be variable and in the elderly it can be delayed. Serum digoxin concentrations, then, may be less reliable for up to several weeks after commencement of digoxin therapy or indeed a change in dosage making assessment of both response to treatment and toxicity in this window difficult. Moreover, low serum concentrations are known to present difficulties in assay. Despite that limitation, the sympatholytic effects of digoxin can be achieved at concentrations well below those required for a positively inotropic effect [26]. In the elderly, increased sensitivity to digoxin may lead to inotropic effects at low concentrations. Furthermore, in older patients (60+ years) dosages based on the Cockcroft-Gault equation do not predict serum concentrations [9]. This, the tool designed to modify digoxin dosage to accommodate the alterations associated with aging, are not reliably informed by serum assays. This reflects the lack of ability of the serum concentration to predict the complex pharmacokinetic and pharmacodynamic behaviour in the elderly.
CONCLUSION
Digoxin remains a widely used pharmacologic agent in clinical practice, and is often prescribed among the elderly patients. Aging is an ongoing process that culminates in structural and functional changes in older populations and these changes have marked affects on critical organ systems. Reduced homeostatic capacity and reduced functional reserve have significant pharmacokinetic implications. Reduced functional reserve increases the vulnerability of older people to disease and drug toxicity. Aging leads to changes in body composition which, combined with decreased renal function and volume of distribution, prolongs the plasma elimination of digoxin and increases bioavailability. Increased risk of pharmacokinetic toxicity is exacerbated by pharmacodynamic increases in digoxin sensitivity. An intimate understanding of digoxin pharmacokinetics, particularly in the older person, will not only improve the quality of prescribing in this cohort, but will also extend advantage to the patient; improved therapeutic outcomes, improved compliance, reduced morbidity and improved quality of life. Specific recommendations for digoxin use in the elderly include:
- minimise the total number of medications,
- discontinue if no benefit is established,
- due consideration to drug-drug interactions and the effects of co-morbidity,
- undertake regular medication reviews and capitalise on therapeutic drug monitoring,
- due consideration for pharmacokinetics,
- employ a dose form that will maximise concordance,
- start doses low and escalate slowly.
While digitalis has been used therapeutically for more than 200 years, ACE inhibitors combined with a diuretic are the current first-line therapy for the management of heart failure. Nonetheless, digoxin has an important role in patients who do not respond to these medications. Digoxin has a particularly important role in older sufferers of heart failure because it reduces the number of hospital admissions associated with escalation of symptoms. This, in conjunction with the increased incidence of heart failure in older people and the aging population, mean the digoxin will continue to be an important and frequently prescribed medication in older people. In the elderly, special consideration and dispensation needs to be applied for digoxin’s narrow therapeutic index, co-morbid diseases, polypharmacy and altered pharmacokinetics to minimise toxicity and sub-optimal therapy.
FUNDING STATEMENT
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.


